The widespread adoption of vaccines over the last two centuries has transformed medicine by preventing infectious disease. In the future, so-called therapeutic vaccines could be used as effective treatments for cancer and autoimmune disorders — ushering in a new era of medicine.
Vaccines are traditionally viewed as a technology that arms the immune system before the body encounters an infectious disease. This preventive approach has helped fight chickenpox, measles, influenza, and other pathogens. Vaccines have also profoundly reduced the incidence of polio, with only 37 confirmed cases reported in 2016 worldwide (1), and they are responsible for the global eradication of smallpox.
Advances in the use of vaccines to prevent infection have radically improved health around the globe, but there is an equally provocative wave of emerging research in developing selective, therapeutic vaccines designed to treat — not just prevent — noninfectious diseases such as cancer and autoimmune disorders. These vaccine-like therapies aim to specifically target tumors, which normally evade the immune system, and to stall autoimmune diseases such as multiple sclerosis (MS) and Type I diabetes — diseases in which the immune system erroneously attacks one’s own tissues. Several recent advances in this area harness the unique capabilities of polymers and other biomaterials.
This article discusses some of the challenges facing therapeutic vaccines for cancer and autoimmune diseases, and the potential of new biomaterial-based technologies that target these diseases. Therapeutic vaccines fit into the broader scheme of immunotherapies that try to correct or enhance the immune system’s behavior; some of the other exciting approaches, such as cell therapy, are highlighted elsewhere in this special section. This is meant as an introductory article; Refs. 2–6 provide more in-depth discussions of biomaterial-based vaccines for infectious disease, cancer, and autoimmune disorders.
The healthy immune response
Vaccines for infectious diseases equip the immune system to fend off safe mimics of a particular pathogen (e.g., protein fragments or a killed virus), enabling the body to quickly recognize and destroy this same pathogen during an actual infection in the future. The body’s pathogen-specific responses are faster after vaccination, because vaccines create immuno-logical memory that cafn last for decades. The immune system must be able to recognize and quickly destroy pathogens with molecular precision, while avoiding attack of self-cells and tissues through a programmed state known as tolerance.
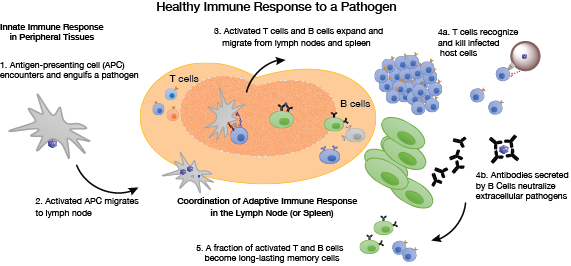
▲Figure 1. A healthy immune system involves a nonspecific innate response that arises quickly but lacks memory and a highly specific adaptive response that requires time to develop initially but creates long-lasting immunological memory. These responses work together to successfully clear pathogens.
Mammalian immune systems have evolved non-specific (i.e., innate) responses that form a first line of defense against foreign invaders, along with highly specific (i.e., adaptive) responses that target distinct pathogens and generate memory against these pathogens (Figure 1).
Innate immune cells — including macrophages and dendritic cells (DCs) — can recognize biological patterns that occur in pathogens and are typically absent in normal human tissues (e.g., lipopolysaccharides that comprise bacterial membranes, DNA, or RNA associated with viruses). Within minutes to hours of detecting the pathogen-associated patterns, innate immune cells mount responses to directly kill the infected cells, engulf pathogens, and release signaling molecules called cytokines to signal danger and recruit more immune cells. These processes underpin the inflammation associated with tissue sites containing infection or wounds.
Innate immune cells do not possess the capacity for memory, so they share information with cells of the adaptive immune system by digesting and presenting the peptide fragments — called antigens — from the digested pathogens. Antigen-presenting cells (APCs), such as macrophages and DCs, present antigens alongside co-stimulatory signals to activate T and B lymphocytes, which are key players of the adaptive immune response. While APCs can encounter lymphocytes throughout the body, APCs eventually migrate to lymph nodes and the spleen to present the antigen to T and B cells residing in these immune tissues. Free antigen can also drain to the lymph nodes and spleen, where it is processed by APCs and then presented to T and B cells.
Each B cell or T cell has a receptor for a specific antigen, which, when activated in the lymph nodes or spleen, only binds with specific antigen molecules — enabling a molecule-specific immune response. The activation process significantly enhances the ability of the B or T cells to recognize a given antigen. Activated B cells mature and secrete antibodies that can neutralize extracellular pathogens or identify pathogens for destruction by other immune mechanisms. T cells have several functions. One type of activated T cell, a cytotoxic T cell, directly seeks out and destroys host cells infected with viruses to prevent the virus from spreading. B cells require other types of activated T cells for activation — demonstrating the interconnectivity between cells in the body’s immune mechanisms.
A fraction of activated T cells and B cells remains in the body after a pathogen is cleared. These persistent cells provide the immune system with memory of the pathogen, enabling lymphocyte populations to have a strong and rapid response to that same pathogen in the future. Generating this protective memory — the goal of most traditional vaccines — is not always efficient or long lasting, which is why multiple immunizations may be required over a short period of time (e.g., hepatitis vaccines) and why adults receive periodic booster shots for certain diseases (e.g., tetanus). References 7–9 provide more detailed information on immune responses.
When the immune system goes wild, or mild
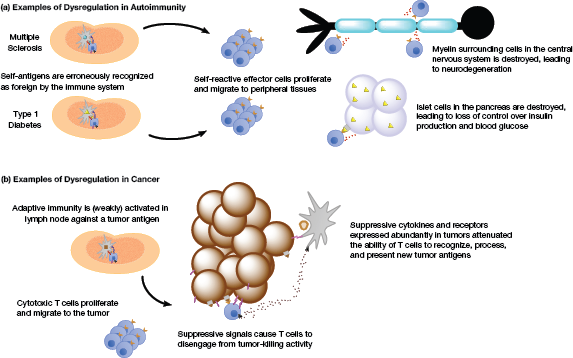
▲Figure 2. Dysregulation of biological signals can cause immune cells to attack one’s own tissues in autoimmune diseases (a) or to ignore unhealthy cancerous tissues that need to be cleared by the immune system (b).
Unfortunately, the normal immune processes can fail — cancer and autoimmune disorders are two pervasive examples. Autoimmune diseases occur when immune cells inappropriately target one’s own cells as if they were foreign or infected (Figure 2a). Some of the most common autoimmune diseases are MS, Type 1 diabetes, rheumatoid arthritis, and lupus. The immune system of someone with MS degrades the myelin sheath around neurons in a process called neurodegeneration. In patients with Type 1 diabetes, the immune system destroys pancreatic islet cells that produce insulin; without enough insulin, the patient’s blood glucose levels are not properly regulated. Therapies that direct the immune system to stop attacking specific “self” molecules — termed tolerance — could stop or reverse autoimmune diseases without causing the broad immune system suppression characteristic of many existing treatments.
Cancer, on the other hand, is a disease in which unhealthy self-cells lose metabolic control and divide in an unrestrained manner (Figure 2b). Cancer cells are able to avoid attack by the immune system by secreting suppressive signaling molecules and presenting those molecules to immune cells. Therefore, even if a T cell is activated against a cancer antigen and infiltrates a tumor, the tumor may signal the T cell to ignore the tumor (tolerance), or the tumor may develop new self-antigens not recognized by the infiltrating immune cells. Moreover, cancer vaccines that generate tumor-specific T cells are risky, because those lymphocytes could recognize noncancerous tissues. Effective cancer vaccines must therefore program the immune system to selectively recognize tumors and overcome the tumor’s immunosuppressive microenvironment.
Regulating the immune system’s overzealous mistakes in autoimmunity and its lack of action toward cancerous cells presents a multitude of opposing challenges. The next section discusses new approaches to tackle cancer and autoimmune diseases with therapeutic vaccines that combine biomaterials with insight gleaned from conventional vaccines against infectious disease.
Biomaterials in conventional vaccines
To generate memory against a particular pathogen, vaccines for infectious disease use attenuated pathogens (e.g., modified by chemicals or heat) or specific antigens isolated or derived from a pathogen. Most vaccine formulations contain antigens along with additives known as adjuvants (10). It has only been in the past few decades that researchers have taken an in-depth look at adjuvant material properties (e.g., particle size, surface charge, specific interactions with innate immune cells) to understand how they influence vaccine performance (11).
Perhaps not surprising then, biomaterial-based...
Would you like to access the complete CEP Article?
No problem. You just have to complete the following steps.
You have completed 0 of 2 steps.
-
Log in
You must be logged in to view this content. Log in now.
-
AIChE Membership
You must be an AIChE member to view this article. Join now.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
