Evaporation is a standard thermally driven unit operation. Learn three methods to improve evaporator efficiency.
Evaporation is a unit operation that separates a liquid from solids by means of heat transfer via vaporization or boiling. The purpose of evaporation is to concentrate a solution of a nonvolatile solute (i.e., solids) and a solvent (i.e., liquid), which is typically water. Evaporating a portion of the solvent concentrates the solute into a more-viscous liquid product. Evaporation is regularly used in the food processing, chemical, kraft paper, and pharmaceutical industries to produce liquid concentrates.
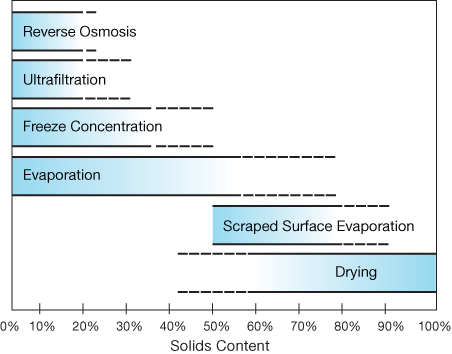
▲Figure 1. Evaporation as a method of concentration can produce solids contents that range from 0% to as much as 92% with scraped surface evaporators (used in select applications).
Evaporation differs from dehydration and drying in that the product of evaporation is a concentrated liquid, not a solid. Evaporation can be used as the initial step in producing a dried product if the liquid concentrate then undergoes a drying process such as spray drying. The combination of evaporation and spray drying is often used to make powdered products, such as powdered milk. This combination of processes is economically attractive because high-efficiency evaporation is significantly less costly than drying and other methods of removing water (1). Evaporation also produces a higher concentration of solids than other methods of concentration (Figure 1) (2).
Evaporation differs from distillation in that the concentrated solution, rather than the condensed evaporate, is typically the valuable product. A common exception is the evaporation of solutions with a high mineral content, where the vapor is condensed as the product and the concentrated brine is discarded. This process is commonly referred to as water distillation, although the process is more akin to a thermally driven liquid-solids separation operation.
Evaporation may be carried out as a batch or continuous process. This article focuses on evaporation as a continuous process, in which the feed and product streams are continuous and their concentrations remain constant.
The basics
All evaporators are comprised of two sections: a heating section (called a steam chest) and a vapor/liquid separation section. These sections can be located within a single vessel (body), or the heating section may be external to the vessel that houses the vapor/liquid separation section.
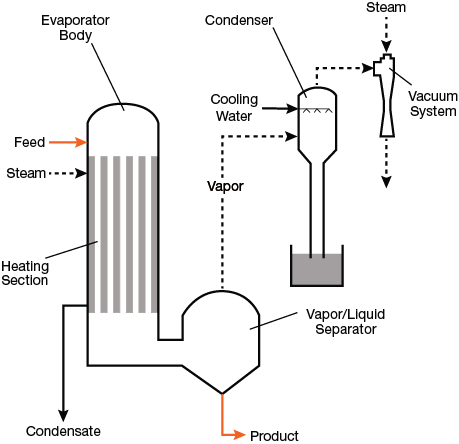
▲Figure 2. The heating and vapor/liquid separation sections are separated in this diagram, but in some types of evaporators, they may be located in the same body.
Evaporators may be composed of one or more effects, where an effect is defined as one or more bodies operating at the same boiling temperature. In a multiple-effect evaporator, vapor from one body heats a second body at a lower boiling temperature. The first effect is heated directly with steam, and the additional bodies are ordered based on descending boiling temperature (or pressure). Figure 2 shows a simple, single-effect evaporation system, which includes an evaporator body with an internal heating section, vapor/liquid separator, water-cooled condenser with barometric leg, and steam-ejector vacuum system.
Evaporator bodies are typically operated under vacuum to reduce the temperature of boiling (e.g., 85°C). Steam ejectors or mechanical vacuum pumps are often used to create a vacuum. Depending on the level of vacuum required for the last effect, which has the lowest boiling temperature, a single pump or a series of pumps may be used.
Vacuum systems also remove noncondensible gases that originate as dissolved gases in the feed or from air leaking into the evaporator body. Most evaporation systems include either a direct or indirect water-cooled condenser to condense the vapor leaving the last evaporator effect. This increases the vacuum of the system. Evaporators that use mechanical vapor recompression (MVR) (discussed later) do not need an external condenser because the vapors generated are fully condensed within the heating section of the evaporator.
Mass balance on a single-effect evaporator


▲Figure 3. An energy balance for this evaporator assumes no work is performed on the system and heat loss is zero. To get a closer approximation of the evaporator rating, however, the heat loss is estimated to be 2% of the vapor energy input to each effect.
Drawing a control volume around the evaporator allows us to perform an energy balance of the system, in which h is the enthalpy of the respective streams:
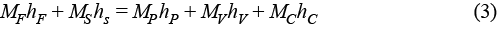
The energy balance assumes that no work is performed on the system (W = 0) and heat loss is negligible (Q = 0). In real applications, heat loss is typically estimated to be 2% of the vapor energy input to each effect (2). Latent heat from saturated steam drives the evaporation of water, and depending on the initial feed temperature, may also be used to raise the temperature to boiling.
The product concentration, where xF and xP are the mass fractions of the respective streams, can be calculated from:

Steam economy
Another way to measure and compare the performance of evaporators is to consider the amount of water evaporated per amount of steam consumed. This metric is called the steam economy (SE) and is commonly expressed as the ratio of water evaporated to steam consumed.
In theory, the evaporation of 1 kg of water requires slightly more than 1 kg of steam, since the latent heat of vaporization decreases as pressure increases (3). As the steam’s latent heat is transferred to the feed, some of the heat raises the initial feed temperature to the boiling point and the remaining latent heat evaporates water. Thus, the steam economy will always be less than one. Depending on the initial feed temperature and system losses, the steam economy of a single-effect evaporator typically is 0.75–0.95 kg water/kg steam (4).
Improving steam economy. Consider the example of the single-effect evaporator (Figure 3). For heat transfer to occur, the temperature of the saturated steam must be higher than the boiling temperature of the feed. If the latent heat of the water vapor leaving the evaporator can be reused, the steam economy can be increased. But, because heat transfer requires a temperature gradient, the latent heat of the water vapor can only be transferred to a lower-temperature fluid.
The two primary means of improving the steam economy of an evaporator both involve manipulating temperature via pressure change.
Multiple-effect evaporators
If the water vapor from the first effect of a multiple-effect evaporator can be introduced into the steam chest of a second effect operating at a lower boiling point, the latent heat in the water vapor can be reused. Lowering the vapor pressure of the second effect relative to the first effect lowers the boiling point of the second effect. This arrangement of reusing vapor latent heat is called multiple-effect evaporation.
Figure 4 shows the mass flows in a three-effect evaporator. The vapor from the first effect (MV1) becomes the heating source for the second effect (MS2). This is possible because the second effect’s boiling temperature has been lowered to 68°C (at 29 kPa), which is less than the saturated temperature of the first effect, 82°C (at 51.8 kPa). The latent heat is reused again as MV2 becomes the heat source of the third effect, where the boiling point temperature has been lowered to 57°C (at 17.5 kPa). The steam economy of multiple-effect evaporators can be estimated by (4):

Evaporators are generally rated based on their evaporation rate — the amount of water they evaporate per hour (e.g., kg/hr, ton/hr). The mass balances for the single-effect evaporator in Figure 3, which include the mass flowrates for feed (MF), product (MP), vapor (MV), steam (MS), and condensate (MC), are:
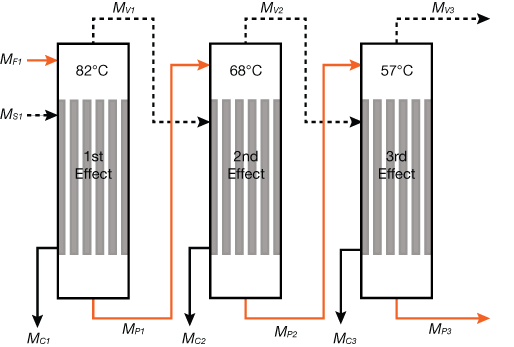
▲Figure 4. The steam economy of an evaporator can be improved by adding more effects. The first effect has the highest boiling temperature, while the last effect has the lowest. Each successive effect uses the vapor from the previous effect as a heat source.
Recompression
The second method for reusing the latent heat of vapor is to increase its condensing temperature by compressing it. This increases the vapor’s condensing pressure and temperature and creates the temperature gradient necessary for latent heat reuse within the evaporator system.
Thermal vapor recompression (TVR) uses a steam ejector to compress the vapor leaving the evaporator vessel. In this application, the steam ejector is often called a thermocompressor; its primary purpose is to compress vapor, but its secondary benefit is creating vacuum within the evaporator vessel. As a thermocompressor, the ejector uses high-pressure steam (0.8–2.1 MPa) as the motive fluid to draw in the low-pressure vapor from the evaporator and produce a combined vapor of suitable pressure and temperature for evaporator heating.
Figure 5 illustrates one possible configuration of a three-effect evaporator with TVR. In this example, the thermocompressor raises the saturation temperature of the vapor from 71°C to 88°C so it can be reused to heat the second effect operating at 80°C.
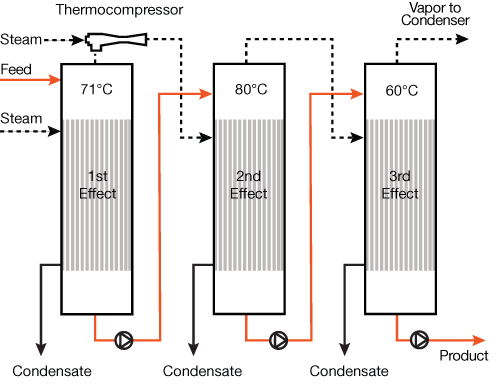
▲Figure 5. Thermal vapor recompression (TVR) can be applied to an evaporator system to improve the steam economy. A thermocompressor raises the saturation temperature of the vapor exiting an effect to be reused to heat the next effect.
Steam ejectors are better suited to handling large volumes of low-density vapor than mechanical vacuum pumps and are easier and cheaper to maintain. As a rule of thumb, TVR improves an evaporator’s steam economy by an amount equivalent to the addition of another effect, but at a much lower cost (5).
Mechanical vapor recompression (MVR) uses a mechanical compressor or fan to compress low-pressure vapor leaving an evaporator to a higher pressure and temperature for reuse as the evaporation heat source. The internal reuse of the vapor also eliminates the need for an external condenser, which most evaporator systems need to condense the vapor leaving the last effect. Because compression ratios of 1.2–1.4 are sufficient to produce a condensing temperature increase of 6–12°C, centrifugal fans are used for their large volumetric capacity, economy of operation, and relatively low cost. Steam turbines are typically used to drive mechanical fans at installations where the cost of steam is relatively low and low-pressure steam can be used for process heating. Otherwise, electric motors with variable-frequency drives (VFDs) are used to drive mechanical fans.
Figure 6 shows a single-effect, vertical long-tube evaporator with MVR. An MVR evaporator requires steam to bring the system up to boiling temperature during startup, but requires very little steam once operating at steady state, with only the work of the compressor adding energy to the system.
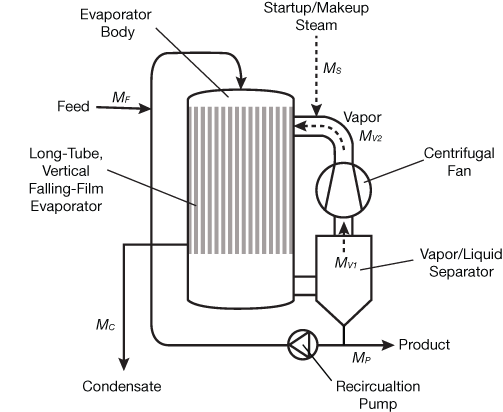
▲Figure 6. Mechanical vapor recompression (MVR) improves steam economy by compressing low-pressure steam exiting the evaporator and recycling it as the heat source.
Comparing steam economies
Table 1 compares the steam economies of multiple-effect, TVR, and MVR evaporators (5). Steam economy increases steadily in multiple-effect evaporators as the number of effects increases. Adding a thermocompressor to a three-effect system can approximately double the economy, but that pales in comparison to the potential steam economy of an evaporator with MVR — which is 5–10 times that of the three-effect evaporator.
| Table 1. Comparison of evaporator steam economies. | |
| Type of Evaporator | Steam Economy, kg water/kg steam |
| Single-effect | 0.90–0.98 |
| Double-effect | 1.7–2.0 |
| Triple-effect | 2.4–2.8 |
| Six-effect | 4.6–4.9 |
| Triple-effect with TVR | 4–8 |
| Single-effect MVR | 10–30 |
Note that the steam economy of an evaporator is highly dependent on the initial temperature of the feed as it enters the evaporator system and the properties of the liquid being concentrated.
Feed that enters the first effect at a temperature above the boiling point first flashes some vapor before heat transfer within the evaporator even begins. Thus, the steam economy will be higher than if the initial feed temperature was below the boiling point, which would require heat transfer to the liquid before evaporation can occur.
Boiling point rise, also called boiling point elevation (BPE), is the difference between a solution’s boiling point and the boiling point of pure water at the same pressure. Some solutions, especially as they become more concentrated, may require a large temperature gradient (16–27°C) to sufficiently transfer enough heat to reach boiling conditions. High temperature gradients limit the number of effects that can be applied, thereby limiting the potential steam economy increase of a system. Also, single-stage thermal and mechanical compressors are economically limited to compression ratios of about two, which produces a temperature increase of 5–20°C (5). Thus, the potential to increase the steam economy by adding effects, TVR, and MVR may be constrained by the thermal and rheological properties of the liquid being concentrated.
Evaporation in industry
Evaporation is used for subtlety different purposes and with different results in each industry. In the food processing industry, food products are concentrated to increase their shelf life, reduce volume and weight, reduce storage costs, reduce transportation costs, and increase value.
Concentration of fresh foods, such as fruit juices, is an economic way to make use of perishable crops during peak harvest periods (6). Fruit juice concentrates and tomato paste are two examples of seasonal foods that evaporation preserves for year-round consumption. Lower water activity, which is a general indication of microbiological stability, preserves concentrated foods.
In the chemical and kraft paper industries, evaporation is often used to recover valuable, nonvolatile chemicals. For example, in the kraft pulping process, water is evaporated from spent cooking liquid (i.e., black liquor), which concentrates the solids. The concentrated inorganic process chemicals can then be refined for reuse within the process.
In the pharmaceutical industry, evaporation is often used to create a concentrated solution, which can then be dried to create a powdered product.
Evaporators in the food industry
The economics of a process dictate the evaporator type and configuration best suited to a particular application. The three most common types of evaporators used in food concentration are long-tube vertical evaporators, forced-circulation evaporators, and plate-type evaporators.
Falling-film evaporators are constructed around a vertical bundle of large-diameter long tubes, with a liquid distributor on top and a vapor/liquid separator section below. Liquid is fed into the top of the heat exchanger tubes and flows as a film down the inner walls. Heat transferred from the condensing steam through the tube walls creates boiling conditions, and the vapor generated escapes into the inner void. As the film flows downward and water evaporates, the film becomes thinner and the liquid becomes more viscous. Since the film flows by gravity down the long tube, residence times in the heat exchanger section are short (0.5–2 min) (3), which is beneficial to heat-sensitive foods, such as fruit juices and milk.
Forced-circulation evaporators use a centrifugal pump to recirculate the liquid through the heat exchanger (i.e., steam chest), where the liquid is heated by steam. As liquid is pumped upward through the heat exchanger, the static head pressure suppresses vapor formation until the heated liquid enters the vapor/liquid separator, where a portion flashes to vapor. The centrifugal pump allows the residence time in the heat exchanger to be controlled based on the heat-sensitivity of the liquid being concentrated. This feature is important when concentrating pulpy, viscous liquids, such as tomato juice and mango juice.
Plate-type evaporators operate similarly to long-tube vertical evaporators, but with the film flowing on plates rather than tubes. Their advantages over tubular-type evaporators include compact size that allows installation within buildings and their ability to be disassembled for easier cleaning.
Wrapping up
The primary means of increasing the steam economy of an evaporator is to reuse the latent heat of the water vapor. A multiple-effect evaporator uses the water vapor from one effect as the heating medium for the next effect, which operates at a lower boiling point. The latent heat in water vapor can also be reused by thermally or mechanically compressing the vapor to a higher pressure and temperature. These three methods of improving efficiency have their limits, however, which are specific to the physical properties of the liquid feed being evaporated.
Literature Cited
- Smith, P. G., “Evaporation and Drying,” Chapter 12 in “Introduction to Food Engineering,” Kluwer Academic/Plenum Publishers, New York, NY (2003).
- Morison, K. R., and R. W. Hartel, “Evaporation and Freeze Concentration,” Chapter 8 in “Handbook of Food Engineering,” 2nd ed., Heldman, D. R., and D. B. Lund, eds., CRC Press, Boca Raton, FL (2007).
- Saravacos, G. D., and A. E. Kostaropoulos, eds., “Handbook of Food Processing Equipment,” Kluwer Academic/Plenum Publishers, New York, NY (2002).
- Chen, C. S., and E. Hernandez, “Design and Performance Evaluation of Evaporation,” Chapter 6 in “Handbook of Food Engineering Practice,” Valentas, K. J., et al., CRC Press, New York, NY (1997).
- Minton, P. E., “Handbook of Evaporation Technology,” Noyes Publications, West Wood, NJ (1986).
- Roa, M. A., and A. A. Vitali, “Fruit Juice Concentration and Preservation,” Chapter 7 in “Handbook of Food Preservation,” Rahman, M. S., ed., Marcel Dekker, New York, NY (1999).

where e is the steam economy of a single-effect (between 0.75 kg water/kg steam and 0.95 kg water/kg steam) and N is the number of effects.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
