As opportunity crude usage increases, unexpected corrosion has become a major issue in piping and process units. Corrosion monitoring is a crucial part of maintaining the operational integrity of a refinery.
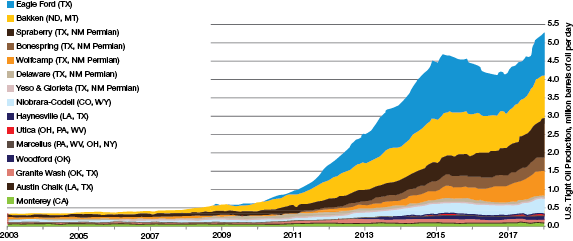
▲Figure 1. The past decade has seen tremendous growth in the amount of tight oil production per day from various plays. Source: Adapted from (1).
In the past five years, there has been significant growth in U.S. production of tight oils, and petroleum refiners are now processing larger quantities of discounted opportunity crude oils than ever before (Figure 1) (1).
Opportunity crude oils are abundant, and their properties can vary significantly. These crudes may have an API gravity that is heavy, intermediate, or light; they can have sulfur content that is higher (more sour) or lower (more sweet); and they can have a higher total acid number (TAN) than traditional crude oils used at a refinery.
Most refineries process many different types of crude, and coastal refineries often process 50 or more different crude oils in a year. Each crude requires slightly different processing conditions within the major process units. However, refinery configurations are typically fixed — they are optimally designed for a specific crude oil composition and desired product qualities and quantities, allowing some flexibility based on the catalyst within the reactors. Therefore, refiners try to match the crude oil composition to their refinery configuration, usually by blending two or more crude oils. Refiners must consider the crude blend’s final properties, as well as whether the blend is incompatible with processing conditions or materials of construction (e.g., whether it may cause accelerated fouling due to precipitating asphaltenes) or has a higher risk of corrosion.

▲Figure 2. High-TAN crudes with high concentrations of naphthenic acids can cause corrosion in piping. Severe corrosion can occur when acids start to form a groove in the bottom of piping, which in turn causes the concentration of acids to increase further in the groove, leading to more corrosion. If the problem is not noticed, the piping can begin to leak.
Corrosion is one of the biggest challenges facing refiners that process opportunity crude oils. Corrosion can create operational challenges, such as unexpected piping leaks, and can necessitate unplanned interruptions and shutdowns (Figure 2). However, not all opportunity crudes cause unexpected corrosion. Many factors can influence corrosion, including the acid types within the crude, distribution of the acid quantity within crude oil boiling point ranges, piping metallurgy, and added inhibitors.
This article explores the types of corrosion that can occur in refineries, techniques to monitor corrosion within piping, and best practices for implementing a corrosion monitoring program.
Types and causes of corrosion
The TAN value is a measurement of acidity in the crude oil. A crude’s TAN value is equal to the amount of potassium hydroxide (KOH, in mg) that is needed to neutralize the acids in one gram of crude oil. High-TAN crudes (crudes with a TAN value of 1.0 or higher) typically have a high naphthenic acid content. It is typically the naphthenic acids in the crude oil that cause corrosion; this type of corrosion is called naphthenic acid corrosion (NAC). Many North American refiners are upgrading their piping to process a higher percentage of high-TAN crude oils.
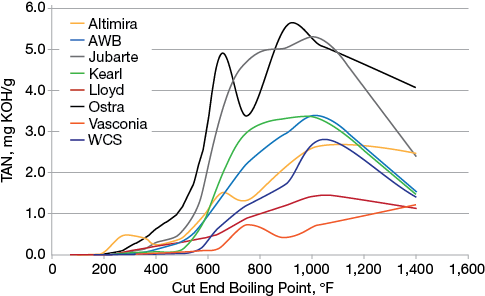
▲Figure 3. A crude oil’s TAN value can vary depending on the cut end boiling point of the process. Source: Adapted from (2).
Although TAN indicates the potential for corrosion, this single number does not indicate what type of acids are present or the naphthenic acid content in the crude. Additionally, the TAN value does not indicate the distribution of these acids within the crude oil’s boiling point ranges (cut points). Acids are not evenly distributed throughout the full boiling point range of the crude oil (Figure 3). Therefore, two crudes with similar TANs can have very different corrosion potential (2). For example, one crude may have the majority of the acid content in the light atmospheric gas oil cut (~500–600°F range) and have a very high corrosivity potential based on the acid type, while another crude oil may have the majority of the acid content in the heavy atmospheric gas oil cut (~600–800°F range) but have a low corrosivity potential because of the acid type.
The TAN measurement alone does not provide all of the insight necessary to mitigate the risk of corrosion. In fact, many types of corrosion can occur at different locations within a refinery, including:
- amine corrosion
- ammonium chloride corrosion
- hydrochloric acid (HCl) corrosion
- naphthenic acid corrosion (NAC)
- high-temperature hydrogen sulfide (H2/H2S) corrosion
- sour water corrosion (acidic)
- ammonium bisulfide corrosion (alkaline sour water)
- hydrofluoric (HF) acid corrosion
- sulfuric acid corrosion.
Depending on the configuration of the refinery, certain types of corrosion are less likely to occur. For example, if there is no alkylation unit within the refinery, there will be no hydrofluoric acid corrosion or sulfuric acid corrosion (because these compounds are only used as catalysts in this particular unit).
Although corrosion can be expected in certain locations throughout the refinery, especially in the crude unit, corrosion can also happen in unexpected places. For example, a low point or dead-leg in a pipe may collect water and provide a favorable environment for...
Would you like to access the complete CEP Article?
No problem. You just have to complete the following steps.
You have completed 0 of 2 steps.
-
Log in
You must be logged in to view this content. Log in now.
-
AIChE Membership
You must be an AIChE member to view this article. Join now.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
