Evaluating adsorbent bed process design and regeneration procedures can provide opportunities for optimization, ultimately reducing operating costs and emissions.
Adsorbent beds, sometimes referred to as process dryers or treaters, use an adsorption mechanism to remove contaminants like water, hydrogen sulfide (H2S), carbon dioxide (CO2), and other polar molecules from process streams. Drying/treating systems are very common in refineries and petrochemical manufacturing facilities and have widespread applications. They are used to dry instrument air, remove sulfur species and CO2 to meet product specifications, remove poisons from reactor feed streams, and remove water and CO2 from cryogenic fractionation trains. The beds contain an adsorptive desiccant, usually an activated alumina, a molecular sieve, or a combination of the two.
The choice of desiccant depends on the size and type of molecule to be removed, as well as the final product specification. Activated aluminas have high loading capacities for more-polar molecules, but they are not as selective as some of the molecular sieves. A 3A molecular sieve, for instance, will adsorb water and ammonia, but will exclude H2S and CO2, which require at least a 4A molecular sieve. Activated aluminas, on the other hand, simply adsorb molecules based on polarity, and the more-polar molecules will displace the less-polar molecules. Manufacturers produce hybrid adsorbents that offer some of the benefits of both activated alumina and molecular sieve, and can tailor some desiccants for a specific application.
In most process plants, adsorbent beds are very reliable and easy to operate, but they do require periodic regeneration. Over time, operating conditions may change and warrant a review of the treater design, operation, and regeneration procedures to improve efficiency. With changes to energy and utility costs and improvements in desiccant technology, treaters that have been in service for a while may benefit from a fresh evaluation.
This article discusses the operation of adsorbent beds and the typical costs associated with regenerating them. Using examples from an ethylene plant, the article identifies methods for reviewing operation and regeneration techniques to reduce operating costs and emissions.
An overview of dryer operation
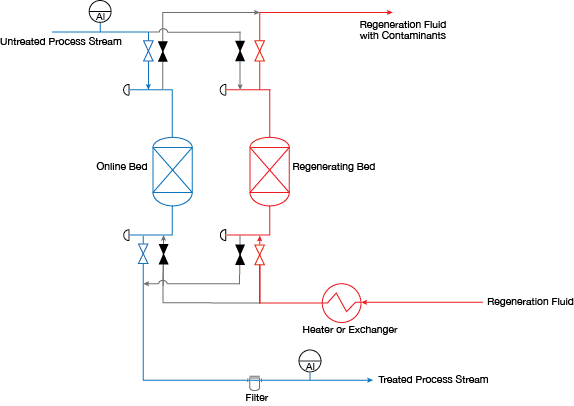
▲Figure 1. In a process dryer/treater system, the process stream is routed through an adsorbent bed. When one adsorbent bed reaches capacity, it is taken offline and the process stream is routed through a second adsorbent bed. Meanwhile, the saturated bed is taken out of service and regenerated.
Regardless of the material or application, process dryers/treaters operate in much the same way. Each process stream is routed through a set of two or more beds (Figure 1). While one bed is in service, the saturated bed is regenerated to remove adsorbed contaminants. Each bed has a fixed adsorption capacity, and once that capacity is reached, the adsorbed contaminants must be removed. The saturated bed is then taken out of service and regenerated.
The regeneration sequence consists of the following five steps:
- Recover the process fluid.
- Heat and purge the bed with a regeneration fluid to shift the equilibrium and remove the contaminants.
- Hold at the elevated temperature to ensure the bed is hot and contaminants are swept from the bed.
- Cool the bed to the process temperature with a contaminant-free stream.
- Return the bed to standby service.
Managing regeneration costs
The long-term costs associated with operating treaters include equipment maintenance and repair and adsorbent replacement. However, the bulk of the operating cost comes from the regeneration cycles. Even the life of the desiccant will depend on the regeneration frequency and regeneration technique employed.
Heating costs and material and disposal costs for the regeneration fluid are the two key costs associated with the regeneration cycle. There are a few ways a facility can reduce the costs of regeneration:
Minimize heating time. The cost to heat the regeneration stream is associated with the steam, electricity, or fuel gas used in a regeneration heater during the heat-up stage of the regeneration cycle. These costs are unavoidable, but can be managed by controlling two variables: first, make sure the regeneration temperature is not excessive, and second, make sure the holding time is no longer than required. Furthermore, reducing the regeneration frequency will also conserve energy, and is discussed in more detail later.
Reduce regeneration fluid costs. Another significant regeneration cost may be the cost of the regeneration fluid. The direct costs associated with once-through regeneration fluids can be high, especially if the fluids are degraded by contaminants and cannot be recovered. The costs incurred to dispose of the regeneration fluid can include, for example, the costs associated with flaring this material.
Plant fuel gas, natural gas, or nitrogen is often used as a once-through regeneration fluid. If the regeneration fluid is fuel gas or natural gas, the spent fluid can be routed internally to the facility’s plant fuel gas system to recover the stream’s heating value, thereby negating the cost of using this material for regeneration.
Unfortunately, plant fuel gas and natural gas may contain some of the same contaminants that the treater beds are designed to remove, chiefly CO2, and so may not be suitable for cooling due to the potential for re-adsorption of these contaminants. This could occur because as a freshly regenerated bed cools, the temperature gradient shifts the equilibrium from desorption to adsorption, and the bed will tend to adsorb contaminants from the regeneration stream. If this is the case, fuel or natural gas may still be a good choice for heating the treater bed during regeneration, but nitrogen may be a better fluid choice in the cooling step because it is free of polar contaminants and is readily available in most facilities.
The challenge with using nitrogen as a regeneration or cooling fluid is disposal cost. Nitrogen that is used for regeneration...
Would you like to access the complete CEP Article?
No problem. You just have to complete the following steps.
You have completed 0 of 2 steps.
-
Log in
You must be logged in to view this content. Log in now.
-
AIChE Membership
You must be an AIChE member to view this article. Join now.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
