Every substance can be toxic, depending on the dose and frequency of exposure. This article outlines the basics of toxicology and explores where the research in this field is heading.
Toxicology is the study of the adverse effects of chemical agents or radiation on living organisms. The field of toxicology has existed since the beginning of recorded history, evolving from the ancient use of poisons for hunting and warfare (1). Today, toxicology is more diverse and progressive, and encompasses exposure and toxicity to humans as well as the environment. It also considers the mechanisms and pathways involved in toxicity and the synergistic effects of exposure to more than one agent.
The concept of the exposome has become a central theme in toxicology. The exposome is a measure of how our environment, diet, lifestyle, and susceptibilities interact and impact our individual response to toxins (2). To complement this, new technologies are able to characterize toxins released into the environment with increasing sensitivity, allowing for more in-depth exposure assessment.
In response to increasing demand for more advanced toxicity assessment, modern toxicology research has started to include high-throughput techniques, such as predictive computer models and automated instruments, to characterize toxicological responses. Researchers are using genomics (i.e., genome analysis) and proteomics (large-scale study of proteins) to study how toxicology is related to the dynamics of an organism. Such techniques produce large data sets that require advanced data management and analysis methods. Furthermore, the demand for high-throughput toxicology research has reduced reliance on animal models and increased the use of synthetic models, such as artificial organs.
Toxicology serves as the basis for regulations that limit hazardous exposures and save lives. For example, the inhalation of dusts generated during coal mining has been correlated with lung disease and death. After standards for coal dust exposure were implemented in the 1970s, the percentage of workers with irreversible lung disease declined.
Unfortunately, there has been a resurgence of irreversible lung disease among coal miners since the early 2000s (3). This is attributed, in part, to a reduction in the size of coal seams and the generation of a dust with higher levels of crystalline silica, which is much more toxic than coal.
This example highlights the importance of implementing conservative protective control strategies to mitigate potentially hazardous exposures, as well as the importance of continuing research on the toxicology of emissions from existing, developing, and evolving processes and products.
Dose and time
A central concept in toxicology is that an agent’s toxicity is defined by the dose (1). In other words, technically, everything is a toxin once the exposure concentration reaches a certain threshold, including water. The relative toxicity of different agents can be compared based on the median lethal dose 50% (LD50), which is the median concentration required to kill 50% of a test population. When extrapolated to humans, water has an estimated LD50 of more than 90 g/kg, whereas alcohol’s LD50 is about 7 g/kg. Information on LD50 and toxic effects are often listed on the safety data sheets supplied by chemical manufacturers.
In addition to dose, time is an important factor, and toxic effects are classified as either acute or chronic. A substance has acute toxicity if adverse effects occur immediately or shortly after exposure. A substance has chronic toxicity if adverse effects appear after a longer period of time, often at a lower dose and after repeated exposures.
For example, hydrogen cyanide gas, which has been used as a chemical weapon, rapidly destroys enzymes that process oxygen, leading to death within seconds of inhalation exposure to a concentrated fume. On the other hand, repeated inhalation exposure to low doses of hydrogen cyanide gas can cause chronic effects such as thyroid enlargement and central nervous system disorders. Chronic effects have been correlated to occupational exposures from the synthesis of polymers and manufacturing activities where hydrogen cyanide gas is a byproduct. Hydrogen cyanide gas also has the potential to affect the general population, as it is found in low doses in smoke from cigarettes, fuel combustion, and burning plastics. Coal miners’ lung disease, discussed in the previous section, is another example of chronic toxicity.
Toxin classification
Agents that can be toxic are broadly categorized as either chemical or radiation. Chemicals are defined as matter in the solid, liquid, or gas phase, ranging from elemental or molecular (e.g., lead, sulfuric acid) to complex mixtures (e.g., diesel exhaust). Radiation is defined as energy in the form of waves or moving subatomic particles, and includes electromagnetic (e.g., ultraviolet, visible light, X-rays), acoustic (e.g., ultrasound, noise), and particle (e.g., alpha particles, beta particles).
Toxin exposure
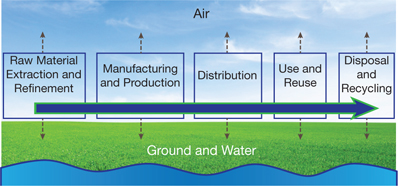
▲Figure 1. Every stage of the product life cycle, from extraction of raw materials through ultimate disposal, can release toxic agents into the environment.
Environmental and human exposure can occur throughout the life cycle of products, from raw material extraction through product disposal (Figure 1). Each stage creates emissions, sometimes in multiple ways.
In the first stage of the product or process life cycle, the mining of raw materials can generate airborne emissions of dusts and acidic gases, and mining wastes and chemicals used in refining can contaminate soil and water. The refining process might involve the handling of materials that could result in worker exposure.
The second stage of the product life cycle — manufacturing and production — can create hazardous wastes that must be properly disposed of to prevent environmental contamination. For example, perfluoroalkyls are frequently used in coatings for products such as carpeting and cardboard packaging. These chemicals have been found to contaminate drinking water near manufacturing facilities. Although studies are still underway, there is evidence that perfluoroalkyl exposure can cause increased blood cholesterol, low birth weight, and liver damage (4).
Distribution of products can create air emissions, via vehicle exhaust. Chronic exposure to toxins in smog can exacerbate lung conditions such as emphysema, bronchitis, and asthma. Exposure to air pollution has also been strongly linked to cardiovascular disease (5).
The use of some products might release additional quantities of hazardous components during the fourth stage of the product life cycle. For example, the chlorine bleach in household cleaning products is a skin irritant, and it reacts with other cleaners, such as ammonia, to produce a poisonous gas that can cause respiratory distress.
Finally, disposal of products may allow hazardous components to leach into the ground and water, or into the air if burned. Hazardous materials in drinking water and air affect humans directly. Agents that accumulate in soil and water affect the heath of wildlife and agriculture and enter the food chain, affecting humans indirectly.
Exposure routes
The most common routes of exposure are through the skin, respiratory tract, and gastrointestinal tract. In addition to causing local irritation and injury, some chemicals can travel from the initial exposure site through blood or lymph vessels to other organs, or produce an immunological response that affects other organ systems.
The skin provides the most significant surface area for potential contamination. Materials absorbed by the skin interact first with the epidermis, which provides a strong physical barrier to protect the underlying dermis. The dermis makes up the majority of the skin and contains a rich supply of blood and lymph vessels, allowing toxins that penetrate the epidermis and migrate to other areas of the body.
When aerosolized materials are inhaled, exposure occurs in the respiratory tract, which includes the conducting and respiratory airways. The conducting airways function to filter, warm, humidify, and transport inhaled air to the respiratory airways, where gas exchange occurs. Most of the conducting airways are protected by a layer of mucus, which traps particles and acts as a diffusion barrier for gaseous species. Just beneath the mucous layer lie cells covered by cilia that beat continuously, transporting mucus and trapped species via the mucociliary escalator to the pharynx, where they are swallowed.
The respiratory airways are protected by immune cells, which engulf and clear foreign matter. The respiratory airway walls are thin to allow for gas exchange, which provides the opportunity for inflammatory molecules and some toxins to escape the airways and enter lung tissue or the circulatory system. Some particles and fibers, such as certain crystalline forms of asbestos, are not efficiently cleared by the protective mechanisms of the respiratory tract and persist at the site of deposition, leading to permanent scarring and, eventually, cancer. Certain toxins have also been shown to activate the autonomic nervous system in the lung, which can directly affect the cardiovascular system. This is thought to play a role in the correlation between exposure to airborne particulate matter in air pollution and cardiovascular disease (5).
When toxic chemicals are ingested, either intentionally or via the mucociliary escalator, they enter the gastrointestinal tract. The gastrointestinal tract is coated with a layer of mucus that allows selective absorption of ingested substances. Toxins are either excreted, absorbed, or persist locally, causing damage and inflammation.
Hazard exposure regulation
With many forms of chemicals and radiation released through various processes, it is important to consider how we track emissions, set standards for safe exposure levels, and enforce regulations. In the U.S., this is done by several regulatory agencies, each of which focuses on a different type of exposure or source of toxicity, including environmental, occupational, food and drugs, and commercial products. Although some agencies perform research related to their respective missions, the government also funds the National Toxicology Program, which includes three organizations assigned to conduct research and develop data to support environmental, occupational, and food and drug regulations.
Air, water, and land quality are regulated by the Environmental Protection Agency (EPA). Founded in 1970, the EPA consolidated many agencies into a single organization that performs research, exposure monitoring, standard setting, and enforcement (6). It is important to note that standard setting and enforcement activities for several environmental programs have been delegated to state agencies. For example, permits for many emission sources are issued at the state level, and each state must develop a plan to comply with ambient exposure standards.
The EPA maintains public environmental databases. Under the National Ambient Air Quality Standards program, it monitors six pollutants — carbon monoxide, lead, nitrogen dioxide, ozone, sulfur dioxide, and particulate matter — at numerous locations throughout North America, and makes the data publicly available (www.epa.gov/airquality/airdata). Another public EPA database is the Toxics Release Inventory (TRI), which reports the release, transfer, and disposal of toxic chemicals from certain facilities.
Safety in the workplace is regulated by the Occupational Safety and Health Administration (OSHA). OSHA was founded in 1970 in response to an increase in work-related accidents and injuries. OSHA establishes exposure standards in the form of an enforceable permissible exposure limit (PEL), which is a time-weighted average for an 8-hr workday in a 40-hr workweek.
The National Institute for Occupational Safety and Health (NIOSH) is responsible for conducting research to support OSHA. NIOSH sets recommended exposure limits (RELs), which are time-weighted average concentrations for up to a 10-hr workday during a 40-hr workweek.
Additional workplace standards include a short-term exposure limit (STEL), which is a time-weighted average over 15 min that should never be exceeded during a workday, and a ceiling limit, which is a maximum peak exposure that should never be exceeded. Both enforceable OSHA and recommended NIOSH exposure limits can be found in the NIOSH Pocket Guide to Chemical Hazards, which is available online (7). The Pocket Guide also includes information regarding toxicity and personal protection measures.
The Food and Drug Administration (FDA) regulates foods, drugs, medical devices, cosmetics, veterinary products, and radiation from electronics. Certain food and drug products require alternative agency support. For example, alcohol is regulated by the Alcohol and Tobacco Tax and Trade Bureau, and agriculture is regulated by the Food Safety and Inspection Service. The safety of consumer products is regulated by the Consumer Product Safety Commission. Extensive information for each of these agencies is available at their respective websites.
Exposure assessment
The OSHA, NIOSH, and EPA publish standard test methods for determining whether chemical concentrations in a workplace or the environment meet the established exposure limits. The two key approaches for exposure assessment are sample collection with offline analysis, and direct-reading sample measurement with real-time analysis. Although not yet prevalent in the standard test methods, real-time analysis is becoming more popular due to advanced sensor technology and reduced instrumentation size and weight that allow for portability.
Chemicals in the solid or liquid phase that deposit onto surfaces can be collected by wipe methods or on the skin using a patch or hand wash. Liquid chemicals can be transferred directly into a vial. Volatile chemicals in the gas phase can be sampled by drawing air through a thermal desorption tube containing a sorbent appropriate for the chemical of interest. Aerosols can be collected using a pump to draw air through a filter medium, the composition of which depends on the desired offline characterization method. A size-selective inlet (e.g., cyclone or impactor) can be installed upstream of the filter to isolate aerosols within specific size ranges. Small aerosols in the submicron size range are more efficiently collected directly onto conductive substrates via electrostatic or thermophoretic deposition.
Chemical identity and concentration are often analyzed offline by chromatography coupled with either a mass, conductivity, or optical-based detector. Aerosols are typically analyzed offline to determine mass, size, morphology, crystallinity, metal content, and carbon content. The mass of an aerosol sample can be measured using a sensitive analytical balance. Solid particles can be counted and analyzed using optical or electron microscopy to obtain data on size distribution and morphology.
Various techniques are often used in tandem — for example, electron microscopes can be equipped with energy or wavelength dispersive X-ray spectrometers, which identify elements based on their unique emission of X-rays upon electron irradiation.
Crystallinity is commonly evaluated by X-ray powder diffraction, which detects the characteristic X-ray diffraction pattern emitted by a crystal structure upon irradiation with X-rays. Metal content is measured by vaporizing the aerosol and determining the mass of individual ions or their electromagnetic properties. Carbon content is commonly determined by combusting a sample to completion and measuring the carbon dioxide formed using an infrared or electrochemical sensor.
There is a broad range of direct-reading techniques available for monitoring an exposure environment in real time. Some instruments are capable of identifying chemical classes and performing elemental analysis based on electromagnetic properties. Portable gas-chromatography/mass-spectrometry (GC/MS) instruments can identify chemicals and in some cases quantify their concentrations. Colorimetric indicator tubes and wipes can be used to identify a single element or compound. Direct-reading instruments, such as photoionization detectors, electrochemical sensors, and metal oxide sensors, can be used to detect different classes of chemicals in real-time. Direct-reading aerosol instruments characterize particle concentration, or both size distribution and particle concentration, in real-time. Particles can be separated by size based on aerodynamic or electrostatic properties, then counted using either an electrometer or laser.
Toxicology research
We are exposed to a broad range of chemicals and radiation in our daily lives; therefore, a considerable amount of research is devoted to understanding the correlation between exposure and toxic effects. In general, results from toxicity studies are used to evaluate potential risks posed to humans as a result of material exposure, as well as to identify and understand the cellular and molecular mechanisms by which they exert toxic effects. Overall, the ultimate goal of toxicity analysis is to support a regulatory framework that protects workers, the general public, and the environment from exposure.
One approach for studying toxicology is to use epidemiology, in which previous exposure and the health of populations are studied to develop relationships between toxic exposures and effects. Because epidemiological and clinical data are rarely available for new materials, additional approaches are used to assess toxicity, including:
- in silico — using software
- in vivo — within a living organism
- ex vivo — in an external environment using tissue from a living organism
- in vitro — in an artificial environment outside a living organism.
Although much of our knowledge about toxicology has come from animal-based studies, in silico and in vitro methods are rapidly becoming more desirable, because they have higher throughput and do not inflict harm on living organisms.
The global in vitro toxicology testing market has been estimated to reach $9.9 billion by 2017, representing a compound annual growth rate of 14.7% from 2012 to 2017 (8). The growth of the market is largely attributed to the high costs, time requirements, and failure rates associated with animal studies (9).
However, current in vitro models have shortcomings, as they are often limited to acute exposures and basic endpoints for cell death, morphology, oxidative stress, and release of inflammatory markers. To further advance in vitro particle toxicity testing, scientists will need to create relevant models that mimic whole organs. More advanced in vitro models are under development, the most promising of which is the microfluidic platform (10). In a microfluidic device, multiple cell types can be incorporated into a well-controlled microenvironment with dimensions and shear rates that correspond to the target organ (11).
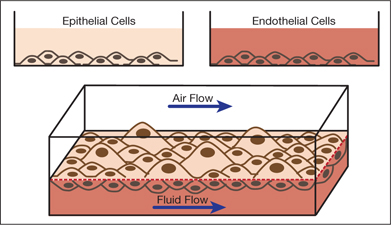
▲Figure 2. In this in vitro model of the air-blood barrier in the lungs, two types of cells — epithelial cells and endothelial cells — are separated by a thin membrane. In the top chamber, air flows past the epithelial cells to mimic breathing. In the bottom chamber, fluid flows past the endothelial cells to mimic blood flow.
Figure 2 illustrates an example of a microfluidic platform — the air-blood barrier where gas exchange occurs. Two types of cells are grown in chambers separated by a thin membrane. In the top chamber, air flows past lung epithelial cells similar to those in the alveolar region of the airways. In the bottom chamber, fluid flows at a rate that produces shear forces on endothelial cells to mimic blood flow. Since it is constructed out of an elastic polymer, the entire device can be cyclically compressed to mimic breathing. Lung cells exposed to physiologically relevant cyclical compression and shear forces have been shown to behave more like those in a whole organ than cells grown in static conditions in a dish (11). Furthermore, studies have shown that devices like these have the ability to mimic whole organ responses to disease and harm (11). This technology is now being commercialized and has fueled the development of a wide range of additional approaches for synthetic biology.
An additional challenge related to toxicology research is investigating the toxicity of particulate matter where the dose applied to the model is not necessarily proportional to the environmental exposure. Particle dosimetry is different from chemical dosimetry in that physicochemical properties influence particle transport (12). Dosimetry is the calculation of the radiation or chemical dose received by the human body.
When assessing particle toxicity, in vitro studies are typically carried out by dispersing particles in cell culture media. During the dispersion step, complex agglomerates consisting of particles and adsorbed media components (such as proteins) are formed. Cell culture media does not have the same properties as bodily fluids, so agglomeration trends will inevitably be different from those in an animal.
In addition to characterizing dosimetry, the exposure route studied should represent realistic conditions. For example, when the lung is exposed to particles via inhalation, the particles are deposited from the gas phase onto cell layers covered in a thin layer of mucus. This is difficult to replicate in in vitro studies. There is extensive research going on in this area, and some progress is being made through dosimetry models and development of complex exposure apparatuses.
Particle size matters
Decades of toxicology and epidemiological research has shown that fine and respirable particles are more hazardous than larger particles (5).
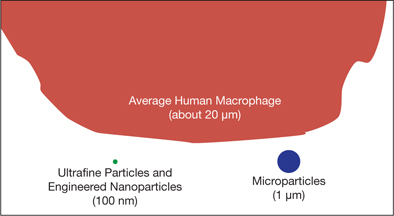
▲Figure 3. Engineered nanoparticles and ultrafine particles can evade many of the body’s natural clearance mechanisms, such as macrophages, due to their small sizes.
Recent evidence suggests that there is an additional size fraction, ultrafine (≤100 nm), that is an exposure concern (Figure 3). Ultrafine particles (UFPs) are emitted during processes in which materials are combusted (e.g., transportation, energy production), heated (e.g., welding), or manipulated with mechanical force (e.g., construction). Studies comparing UFPs to fine particles have shown that toxicity correlates better with particle dose expressed as surface area than as mass when the particles have the same composition (13, 14).
Engineered nanoparticles are an emerging class of materials that have a size range similar to that of UFPs. Nanoparticles are synthetically produced for a broad range of applications and products. The government has invested almost $21 billion in nanotechnology since 2001 (15). The number of products incorporating some form of nanotechnology has been rapidly growing. In 2013, more than 1,600 commercial products were recorded in the Nanotechnology Consumer Products Inventory (16). The inventory is a collection of consumer products reported to contain nanoparticles by the manufacturer or a third-party source. It is important to note that companies are not required to report the use of nanoparticles in their products, and products may claim to incorporate nanotechnology even if their nanoparticle content has not been scientifically verified. Thus, the inventory serves only as a qualitative estimate of the actual number of nano-products on the market.
Nanoparticles and ultrafine particles have caused concern because they can be deposited throughout the respiratory tract and migrate to other organs. Nano-sized particles deposited on the nasal membrane have been shown to move through the olfactory nerves to the brain (17). The role of macrophages (or phagocytic cells, i.e., cells that ingest foreign particles) is to internalize foreign particles deposited in the airways and clear them via the mucociliary escalator. However, UFPs deposited in the airways have been shown to diffuse into the interstitial space or blood circulation, indicating that they can escape the body’s natural clearance mechanisms (18). Studies have also found that UFPs can be internalized by nonphagocytic cells and trapped in intracellular structures (19).
With increasing growth in nanotechnology, the risk for exposure to nanoparticles is increasing as well. Proactive toxicology investigations are essential for controlling exposure and mitigating potential negative health impacts. Because the applications are far-reaching, nanotoxicology is relevant to all sectors of industry, including environmental, occupational, food and drug, and consumer products.
Progress in nanotoxicology has been made over the past decade or so. Nonetheless, enforceable exposure standards have not been established, and it is still unclear how to report nanoparticle concentrations — e.g., on a traditional mass basis as is often used for exposure standards, or on a number or surface area basis, which may better describe these tiny particles. Hopefully, advances in toxicology testing will expedite research and enable us to prevent harmful exposures. This is an ongoing challenge.
Final thoughts
Despite years of research and continuously improving technologies, there are still many questions within the field of toxicology. In order to address these questions, many disciplines must collaborate. And although toxicology may not seem relevant to engineering at first, engineers — particularly chemical engineers — are essential.
For instance, engineers working in an industrial environment may be required to help mitigate potentially hazardous exposures and meet exposure limits set by regulatory agencies — e.g., by designing appropriate ventilation systems and personal protective measures. Research engineers can expand our understanding of toxicology, e.g.?, by characterizing the transport of toxic agents and designing instrumentation and test methods that mimic realistic exposure environments. Furthermore, as more complex in vitro models are developed, engineers are required for manufacturing and transitioning these models into practical applications. With this in mind, it is conceivable that many of the approaches to solve research gaps in toxicology lie in the creative mind of a chemical engineer.
CHRISTIN GRABINSKI, PhD, is a research scientist at the U.S. Air Force Research Laboratory (AFRL) (Email: christin.grabinski.1.ctr@us.af.mil) and is focused on characterizing aerosol emissions and transport in Air Force environments where exposure is a concern. She received her PhD at Case Western Reserve Univ. (CWRU) in 2015, performing research under the direction of R. Mohan Sankaran at CWRU and Saber M. Hussain at the AFRL on the design of exposure devices for assessing the toxicity of aerosolized nanomaterials. Grabinski has presented research at 22 local and international conferences and is author/coauthor of 13 peer-reviewed publications, two book chapters, and one patent application. She is a member of AIChE, American Chemical Society, and American Association for Aerosol Research.
Literature Cited
- Klaassen, C., “Casarett & Doull’s Toxicology: The Basic Science of Poisons,” 8th ed., McGraw-Hill Education, New York, NY (2013).
- National Institute for Occupational Safety and Health, “Exposome and Exposomics,” www.cdc.gov/niosh/topics/exposome (2014).
- Blackley, D. J., et al., “Resurgence of a Debilitating and Entirely Preventable Respiratory Disease among Working Coal Miners,” American Journal of Respiratory and Critical Care Medicine, 190 (6), pp. 708–709 (2014).
- Agency for Toxic Substances and Disease Registry, “Toxic Substances Portal: Public Health Statement for Perfluoroalkyls,” www.atsdr.cdc.gov/phs/phs.asp?id=1115&tid=237 (2015).
- Brook, R. D., et al., “Particulate Matter Air Pollution and Cardiovascular Disease: An Update to the Scientific Statement from the American Heart Association,” Circulation,121, pp. 2331–2378 (2010).
- U.S. Environmental Protection Agency, “About EPA: EPA History,” www2.epa.gov/aboutepa/epa-history (2015).
- National Institute for Occupational Safety and Health, “NIOSH Pocket Guide to Chemical Hazards,” www.cdc.gov/niosh/npg (2015).
- BBC Research, “In Vitro Toxicity Testing: Technologies and Global Markets,” Report Code: PHM017E, www.bccresearch.com/market-research/pharmaceuticals/in-vitro-toxicity-phm017e.html (Jan. 2014).
- Hartung, T., “Food for Thought: Look Back in Anger — What Clinical Studies Tell Us about Preclinical Work,” Alternatives to Animal Experimentation,30 (3), p. 275 (March 2013).
- Huh, D., et al., “Reconstituting Organ-Level Lung Functions on a Chip,” Science,328 (5986), pp. 1662–1668 (June 2010).
- Sivagnanam, V., and M. A. M. Gijs, “Exploring Living Multicellular Organisms, Organs, and Tissues using Microfluidic Systems,” Chemical Reviews,113 (5), pp. 3214–3274 (2013).
- Teeguarden, J. G., et al., “Particokinetics In Vitro: Dosimetry Considerations for In Vitro Nanoparticle Toxicity Assessments,” Toxicological Sciences,95 (2), pp. 300–312 (Feb. 2007).
- Oberdörster, G., et al., “Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles,” Environmental Health Perspectives,113 (7), pp. 823–839 (July 2005).
- Donaldson, K., et al., “Concordance Between In Vitro and In Vivo Dosimetry in the Proinflammatory Effects of Low-Toxicity Low-Solubility Particles: The Key Role of the Proximal Alveolar Region,” Inhalation Toxicology,20 (1), pp. 53–62 (2008).
- National Nanotechnology Initiative, “National Nanotechnology Initiative Budget,” www.nano.gov/about-nni/what/funding (Accessed on Jan. 19, 2015).
- The Project on Emerging Nanotechnologies, “Consumer Products Inventory,” www.nanotechproject.org/cpi (2014).
- Elder, A., et al., “Translocation of Inhaled Ultrafine Manganese Oxide Particles to the Central Nervous System,” Environmental Health Perspectives, 114 (8), pp. 1172–1178 (Aug. 2006).
- Nemmar, A., et al., “Passage of Inhaled Particles into Blood Circulation in Humans,” Circulation,105 (4), pp. 411–414 (Jan. 2002).
- Grabinski, C. M., et al., “Multi-Functionalized Spions for Nuclear Targeting: Cell Uptake and Gene Expression,” NANO,9 (1), (2014).

Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
