Apply the fundamental principles of continuous process design and process intensification to change the pharmaceutical manufacturing status quo.
Process intensification (PI) in the pharmaceutical manufacturing industry has been demonstrated in bioprocessing and small-molecule drug facilities through the implementation of continuous manufacturing (CM). Some of the major benefits of CM include faster speed to market, better process control, smaller factories and environmental footprints, more consistent product quality, modular manufacturing, and lower capital and operating costs (1–3).
Although the product design, chemistry, biology, drug delivery method, and efficacy of the drugs are defined by chemists, biologists, and medical experts, equipment and process design are the domain of chemical engineers. Chemical engineers have the skills and tools to design efficient and robust processes to overcome the technical and economic barriers to CM.
Because the technoeconomic criteria, products, steps, and scales vary between applications, each CM process design should be tailored to the specific application. CM processes can be fully end-to-end continuous, a hybrid of batch and continuous, or individual continuous operations retrofitted in a batch process (mostly for legacy processes). Selection of the approach is guided by an evaluation of the product, process, equipment, regulations, enterprise readiness, technology availability, market demand, and volume and/or value of the product.
Pharmaceutical manufacturing is a broad industry in which CM can be applied to different domains. Figure 1 indicates relevant CM methods that can be applied to discrete domains, but domains can be connected in an end-to-end fashion to create a fully continuous process. For instance, synthesis and purification (drug substance), formulation and blending (drug product), and tablet production can all be combined in one train (2).
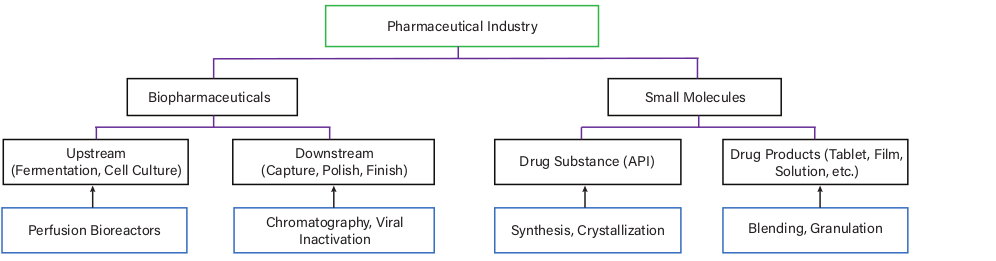
▲Figure 1. These continuous processing methodology examples (blue boxes) can be applied to several different domains within the pharmaceutical manufacturing industry.
Would you like to access the complete CEP Article?
No problem. You just have to complete the following steps.
You have completed 0 of 2 steps.
-
Log in
You must be logged in to view this content. Log in now.
-
AIChE Membership
You must be an AIChE member to view this article. Join now.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
