Versatility, ease of manufacture, and relatively low cost make plastics some of the most useful materials for a wide range of applications. This article explains the chemistry and production processes behind two of the most popular plastics — polyethylene and polypropylene.
Plastics are some of the world’s most diverse and useful manufacturing materials. While plastics encompass a large number of materials, polyethylene and polypropylene are two of the major plastic types found in many consumer goods, from car parts to shopping bags to water pipes.
Several types of reactors can make polymers, including fluidized-bed, loop, autoclave, and tubular reactors. Different grades of polyethylene and polypropylene offer a wide range of physical properties, such as density, stiffness, flexibility, opacity, melting point, texture, and strength. Manipulating variables in the reactor, such as monomer, comonomer, catalyst, and cooling media flowrates, can control key quality parameters. Additives and colorings can modify the appearance of the polymer.
Polymer plants are semi-continuous processes. Raw material is fed into a reactor continuously at the front end, while polymer powder and pellets are packaged in batches. Most sites operate multiple lines with many bins and silos for storage and blending. The plastics are eventually delivered to customers using barges, trucks, or rail cars.
This article describes the different polyolefin production processes, their key operating parameters, and ways to use automation for improved quality control and higher throughput.
The chemistry of making plastic
It’s helpful to understand some of the chemistry behind the polymerization reaction to appreciate how the process works and the complexity involved in making plastic. A polymerization reaction starts with a primary ingredient (monomer), such as ethylene or propylene.
Ethylene (C2H4) is a stable molecule with two carbon atoms and a double bond. Polyethylene (PE) is a made by the reaction of multiple ethylene molecules in the presence of catalyst to break the double bond and connect the carbon atoms into a chain (Figure 1). The longer the chain, the higher the molecular weight. Polymers can have molecular weights in the millions.
Similarly, polypropylene (PP) is made by breaking the double bond in a propylene (C3H6) molecule, in the presence of a catalyst, to form long chains of three-carbon-atom molecules (Figure 2). The third carbon atom adds a complexity: On which side of the chain will the methyl (CH3) groups fall? They could all be on one side of the chain’s centerline, or backbone (isotactic), they could appear alternately on opposite sides of the backbone (syndiotactic), or their positions could be random (atactic). These arrangements have different physical properties.
Polymerization reactions will also consume hydrogen, which is required to quench the reaction (i.e., end the chains), and some will involve a secondary ingredient (known as a comonomer). Since the concentrations of these components in the reactor affect the probabilities that specific reactions will take place, the composition in the reactor effectively sets the amount of branching and the length of the chain.
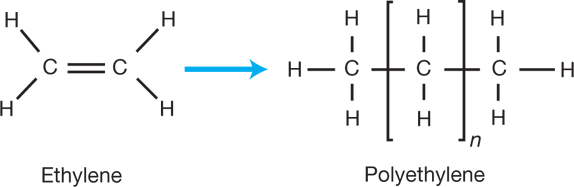
▲Figure 1. Ethylene is a stable molecule with two carbon atoms connected by a double bond. Polyethylene is made by the reaction of multiple ethylene molecules in the presence of catalyst.
The polymer industry employs many catalysts, and new catalysts are developed every year. Different catalysts are used to create polymers with particular properties, even in the same reactor. Each PE or PP process licensor incorporates proprietary catalyst formulations in its reactor designs. Depending on the type of reactor, the catalysts can be solid particles or suspended in hydrocarbon or solvent.
Polymerization is a highly exothermic reaction, and it requires continuous cooling to prevent runaway reactions. Most reactor systems include an emergency quench that quickly shuts down the reactor if the temperature reaches a predetermined setpoint. Before the implementation of redundant control mechanisms, a runaway reaction could cause the reactor to become completely plugged with plastic. Process designs have since changed and safety systems have been implemented to prevent such incidents.
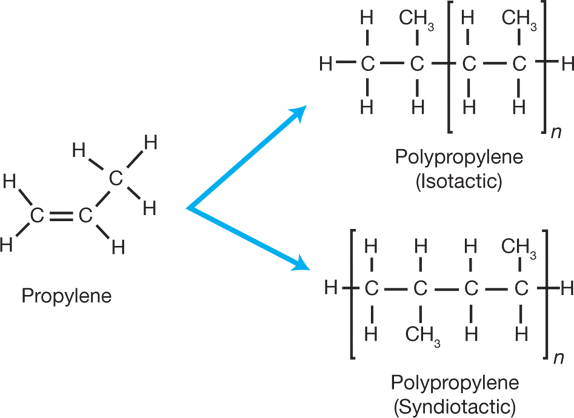
▲Figure 2. Polypropylene is made by breaking the double bond in a propylene molecule in the presence of a catalyst. The resulting polymer may be isotactic, with all of the methyl groups on the same side of the polymer backbone, syndiotactic, with alternate methyl groups on opposite sides of the backbone, or atactic (not shown), with the methyl groups arranged randomly.
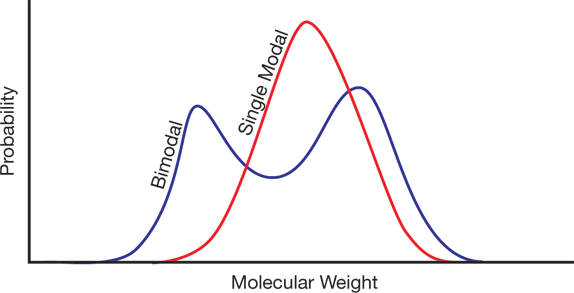
▲Figure 3. The molecular weight distribution of a bimodal polymer has more than one peak.
Key quality specifications
Although polymer properties can be modified somewhat in the blending and extruding steps, the conditions in the reactor set the product grade(s) — as defined by a few key quality measures — that can be made. In general, polyethylene is categorized based on its density, the linearity of the molecules (i.e., degree of branching), and its molecular weight (length of the chains). Other polymer qualities, including the molten flowing property, known as the melt-flow index, are a function of the crystalline structure and are also primarily determined by the polymerization reaction. The melt-flow index determines the polymer’s behavior in downstream operations, such as extrusion, blow molding, or film production.
The most common polyethylene grades are:
- high-density polyethylene (HDPE). This polymer has a density greater than or equal to 0.941 g/cm3. It has a low degree of branching, with mostly linear molecules, has high tensile strength, is resistant to many chemicals, and is used in such products as bottles, jugs, water pipes, and toys.
- medium-density polyethylene (MDPE). With a density range between 0.925 and 0.940 g/cm3, MDPE has better shock and stress crack resistance than HDPE and is commonly used for gas pipes, plastic bags, and packaging films.
- low-density polyethylene (LDPE). This grade has a density range of...
Would you like to access the complete CEP Article?
No problem. You just have to complete the following steps.
You have completed 0 of 2 steps.
-
Log in
You must be logged in to view this content. Log in now.
-
AIChE Membership
You must be an AIChE member to view this article. Join now.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
