Many low-temperature processes leverage nitrogen’s cooling and freezing capabilities. Find out what methods are used in what applications and why.
The chemical process industries (CPI) employ nitrogen — as a gas or liquid — in a wide range of applications (1, 2). Gaseous nitrogen (GAN) can inert vessels and purge lines to eliminate explosion hazards and prevent undesired oxidation reactions that can reduce product quality. Liquid nitrogen (LIN) is used in innovative cooling and freezing technologies.
LIN is an effective and convenient refrigerant due to its availability, low cost, and inert properties. It is also a practical cryogen for most low-temperature applications because of its extremely low boiling temperature (–195.8°C) and high refrigeration capacity at atmospheric pressure. Even at elevated pressures, the thermal properties of LIN (Table 1) make it an effective refrigeration medium to rapidly cool processes to low temperatures.
This article describes methods for employing liquid and gaseous nitrogen’s cooling and freezing capabilities, as well as some specific applications in the chemical and pharmaceutical industries.
LIN refrigeration methods
Several cooling techniques take advantage of LIN’s refrigeration capabilities in batch or continuous processes.
- direct surface (semi-indirect) cooling (Figure 1a). LIN provides cooling via a single conductive wall, the cold surface of which freezes or cools liquid or gas streams.
- secondary circuit (indirect)cooling (Figure 1b). The boiling temperature of LIN is buffered with an intermediate heat-transfer fluid (HTF) to enhance temperature control. The HTF temperature can be tuned to the desired process temperature, as low as the boiling point of LIN. The HTF then provides refrigeration through a conductive wall to freeze materials or cool fluids.
- cold GAN cooling (Figure 1c). LIN vaporizes, and the sensible heat capacity of the cold GAN is used for refrigeration. Additional LIN is injected to control the temperature. Cooling occurs via a conductive surface, or by blowing the cold GAN directly onto the materials to be cooled.
- direct LIN injection/spray cooling (Figure 1d). LIN is injected or sprayed directly onto materials or into processes. Materials and processes are cooled by the latent heat of vaporization of LIN; depending on the design of the cooling system, the sensible heat capacity of the cold GAN may also contribute to the cooling. This is an efficient use of LIN’s refrigeration value.
- immersion cooling (Figure 1e). Direct immersion in LIN cools or freezes a material. The rate of cooling depends almost entirely on the latent heat of vaporization of LIN. The overall heat-transfer rate is generally lower than that of direct LIN injection/spray cooling, because the GAN has a blanketing effect — the bubbles generated by the turbulent boiling of the LIN create a vapor boundary layer around the immersed material, thereby lowering the overall heat-transfer coefficient.
| Pressure, atm | Boiling Temperature (Tsat), °C | Heat of Vaporization, kJ/kg | Liquid Thermal Conductivity, J/m-s-°C | Heat Capacity at Tsat, kJ/kg-°C | Vapor Thermal Conductivity, J/m-s-°C |
| 1 | –195.8 | 5,592.8 | 0.137 | 1.08 | 0.007 |
| 3 | –185.1 | 5,157.5 | 0.118 | 1.15 | 0.009 |
| 6 | –176.6 | 4,720.3 | 0.102 | 1.16 | 0.011 |
| 9 | –170.8 | 4,351.5 | 0.091 | 1.38 | 0.012 |
| 12 | –166.3 | 4,005.4 | 0.083 | 1.53 | 0.013 |
The suitability of a particular cooling method depends on the application and the nature of the materials or processes to be cooled. Cold GAN cooling, for example, is an attractive option for cooling delicate materials, whereas direct LIN injection/spray or immersion cooling may damage the structure of the material. Additionally, cold GAN and secondary-circuit cooling can be employed when it is critical to operate above the freezing point of materials or fluids to avoid freezing that could damage the material or plug the process. Applications where flash freezing is desired often use direct LIN injection/spray cooling or immersion cooling.
These LIN freezing and cooling methods are deployed in a variety of applications. The remainder of this article provides an overview of some LIN cooling/freezing applications in the chemical and pharmaceutical industries.
Cryogenic milling and grinding
LIN refrigeration enables cryogenic milling and grinding to pulverize, to micron- or submicron-sized powders, materials that would otherwise be difficult to mill at ambient temperature. The process is suitable for materials that have high viscoelasticity, adhesive properties, or thermal sensitivity.
Viscoelasticity. Materials like rubber and elastomeric gels have high viscoelasticity — i.e., they resist shear flow, stretch when impacted, and return to their original state when stress is removed. Viscoelastic properties are directly proportional to temperature; therefore, lowering the temperature embrittles the material, which makes milling easier and more effective.
Adhesive properties. Sticky materials like waxes and oily biological samples have a tendency to cling to other materials and surfaces. When milled at ambient conditions, they build up in the mill, thereby lowering the mill throughput, spiking power consumption, and eventually plugging the process. Low temperatures inhibit the adhesion mechanisms responsible for stickiness and increase cohesive intermolecular forces, making the materials more brittle and less sticky and thus easier to mill.
Thermal sensitivity. Some materials lose their chemical, biological, or electrochemical activity at elevated temperatures; for example, heat may damage protein-based pharmaceuticals. Because milling processes generate heat, refrigeration is needed to control the temperature when processing these sensitive materials.
The heat generated by milling also makes it more difficult to process viscoelastic and adhesive materials. Therefore, it is critical to control the temperature of the mill as well as the temperature of the material being milled. Cryogenic milling involves cooling the materials and/or the mill, generally through direct LIN injection (Figure 2). The injected LIN also inerts the atmosphere, which prevents undesired oxidation reactions.
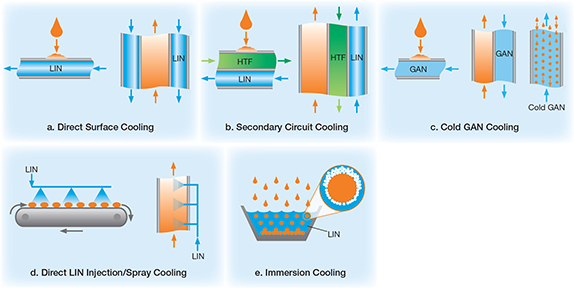
▲Figure 1. LIN refrigeration methods. (a) A conductive wall, cooled by LIN, freezes or cools fluids that contact it. (b) A heat-transfer fluid (HTF) serves as an intermediate cooling agent between the LIN and fluid to enhance temperature control. (c) Cold GAN cools through a conductive wall or can be directly blown onto the fluid to be cooled. (d) LIN is directly injected or sprayed onto a material or into a process. (e) When a material is directly immersed in LIN, bubbles generated from turbulent boiling create a vapor boundary around the material, which lowers the overall heat-transfer coefficient.
Lyophilization
Lyophilization, or freeze-drying, dehydrates thermally sensitive materials (e.g., proteins) by freezing the material with LIN, followed by controlled sublimation under vacuum. Materials may be freeze-dried to preserve microscopic structures (e.g., cell structures) and to enhance product stability for prolonged storage and transportation. Mechanical refrigeration by compressors has been used for freeze-drying applications, but LIN has gained popularity because it enables better control of the freezing process and a broader range of operating parameters (3).
In small-scale operations, materials may be frozen through LIN immersion or direct surface cooling prior to being placed in a vacuum chamber. In larger-scale operations (Figure 3), LIN secondary circuit cooling (using a heat-transfer fluid) is the most economic and effective method of freezing, as it offers quick, cryogenic-temperature freezing and flexible temperature control to speed up vacuum drying. After freezing, a vacuum pump causes sublimation of most of the frozen solvent in the primary drying step. A cryogenic condenser, cooled via LIN direct surface cooling, maximizes the rate of mass transfer of solvent from the frozen material to the condenser surface. Any physicochemically bound solvent (e.g., water) not removed during primary drying is removed during secondary drying, where the material temperature and vacuum are increased. If the material being freeze-dried is oxygen-sensitive, an inert gas backfill breaks the vacuum while maintaining an inert atmosphere.
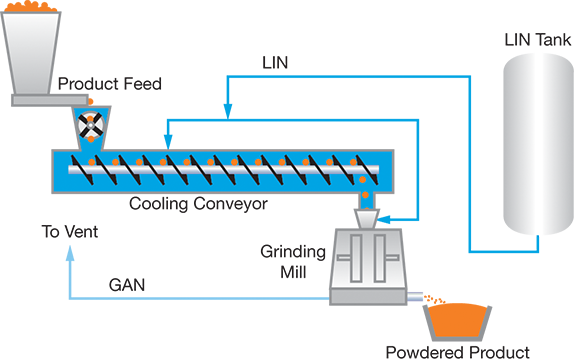
▲Figure 2. In cryogenic milling, LIN cools materials that are difficult to mill at ambient temperature. Because the milling process generates heat, the temperature of the mill is also regulated with direct LIN injection.
Cryopelletization
Cryopelletization is a low-temperature technique for manufacturing pellets that are most often spherical or semi-spherical and range from 0.5 mm to 5 mm in diameter (4). It is often used to pelletize heat-sensitive materials, such as bacterial cultures and probiotics. Because cryopelletization effectively flash-freezes materials, it is used to prevent phase separation of components in solutions, colloids, or suspensions, as well as to form pellets with a high degree of homogeneity. The three common forms of LIN-cooled cryopelletization are direct surface freezing, LIN immersion freezing, and cold GAN freezing.
Direct surface freezing (Figure 4a). Droplets freeze on a cold surface, such as a rotating stainless steel drum that is cooled by direct LIN injection inside of the drum. The pellets formed are consistently semi-spherical with a flat base. The size and shape can be modified by adjusting process variables, such as the distance the droplets fall and the rate of freezing.
LIN immersion freezing (Figure 4b). Droplets freeze as they are immersed in LIN. As droplets are immersed, the LIN boils turbulently, which makes it more difficult to control the size distribution and the shape of pellets. Nevertheless, this method generally produces a high percentage of spherical pellets.
Cold GAN freezing (Figure 4c). Cold GAN freezes droplets as they free-fall, producing spherical pellets with a narrow size distribution. However, it is critical to control process variables, such as droplet size, GAN temperature, and freezing chamber geometry, to prevent droplets from agglomerating or sticking to sidewalls as they fall.
Pelletization is advantageous because it improves product flowability and blendability. Enhanced flowability makes downstream conveying and packaging easier. Pellets can be easily and accurately blended or mixed to form products with a variety of properties. Additionally, pelletization minimizes the creation of fines and dust, reducing the risk of dust explosions and respiratory health effects.
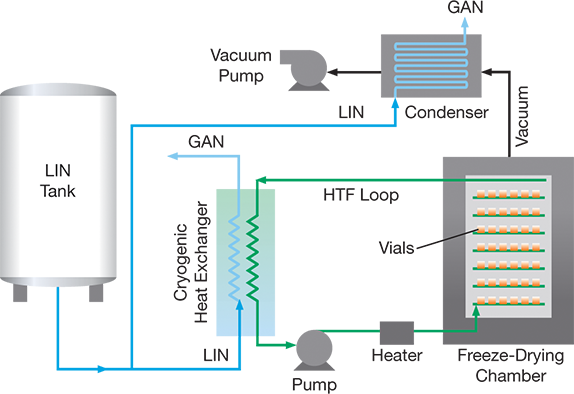
▲Figure 3. LIN provides refrigeration to vials of sample via a secondary heat-transfer fluid (HTF) circuit. During vacuum drying, LIN also cools the condenser to create a low-temperature cold trap for solvent removal.
Cryogenic spray freeze-drying
Cryogenic spray freeze-drying (SFD) creates fine (usually smaller than 100 µm), low-density, dry powders (e.g., inhalable pharmaceuticals) at low temperature. It combines the principles of lyophilization and spray drying.
Fine droplets are created from a solution, suspension, or colloid, and then rapidly frozen via direct LIN spraying (Figure 5a) or LIN immersion (Figure 5b). Single-fluid or two-fluid atomizing and ultrasonic piezoelectric nozzles create the micro- or nano-sized droplets. The liquid-product formulation, atomizing parameters, and freezing rate can be modified to control dry particle characteristics, such as shape, size, size distribution, and overall morphology.
The frozen powder may be the final product, but typically a gentle drying process, such as vacuum (lyophilization) or atmospheric drying, follows the freezing step. Atmospheric drying introduces temperature-controlled, cold, dry GAN to dry the frozen powder through convective mass transfer under atmospheric pressure. The moist, cold GAN is then vented from the process or dehumidified and recycled.
The dry powder particles produced by SFD are spherical, light, and highly porous, and they have attractive aerodynamic properties, making them well-suited for inhalable powdered drugs that are administered through the nasal or pulmonary pathways. Additionally, the dry powders can be reconstituted faster than materials dried by traditional lyophilization and spray drying methods, which is important for pharmaceuticals with low water solubility.
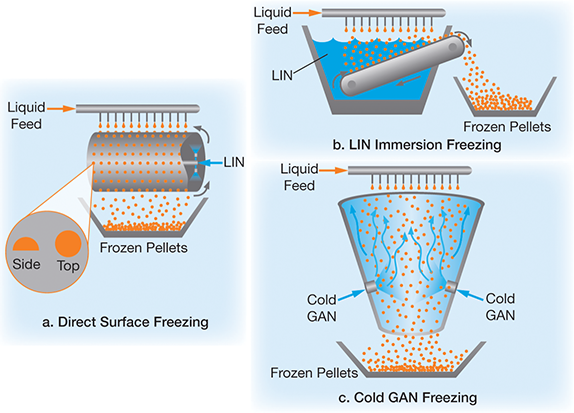
▲Figure 4. Cryopelletization can employ (a) direct surface freezing, (b) LIN immersion freezing, or (c) cold GAN freezing.
Cryogenic thin-film freezing
Cryogenic thin-film freezing (TFF) is a relatively new low-temperature process that produces highly porous, micron- and submicron-sized dry particles of heat-sensitive materials, such as proteins, that are suitable for pulmonary and parenteral drug-delivery applications. The process is similar to cryogenic pelletization by direct surface cooling using a rotating stainless steel drum. Drops of a solution, suspension, or colloid fall onto a cold stainless steel surface from a distance that allows the droplets to collapse into flat, thin (micron- or submicron-thick) films as they rapidly freeze (Figure 6). The film thickness can be modified by adjusting the free-fall distance and the physical properties of the liquid feed. The frozen films are freeze-dried to produce dry powders.
In TFF processing, the gas-liquid interface is minimized during freezing (in contrast to SFD), which reduces protein denaturation (5). TFF is used at the laboratory scale, especially for research on the reconstitution of insoluble drugs. It is not clear whether it will be a cost-effective solution for large-scale manufacturing.
Cryogenic reaction cooling
Cryogenic reaction cooling provides refrigeration and low-temperature control for temperature-sensitive processes, such as highly exothermic and cold-chemistry reactions. For example, in organic and organometallic synthesis, low-temperature operation is important to balance reactivity, selectivity, and yield. For highly exothermic reactions, cooling is critical to control heat release and avoid runaway reactions. Direct injection cooling, semi-indirect cooling, and indirect cooling with HTF are the three primary options for cooling reaction vessels with LIN.
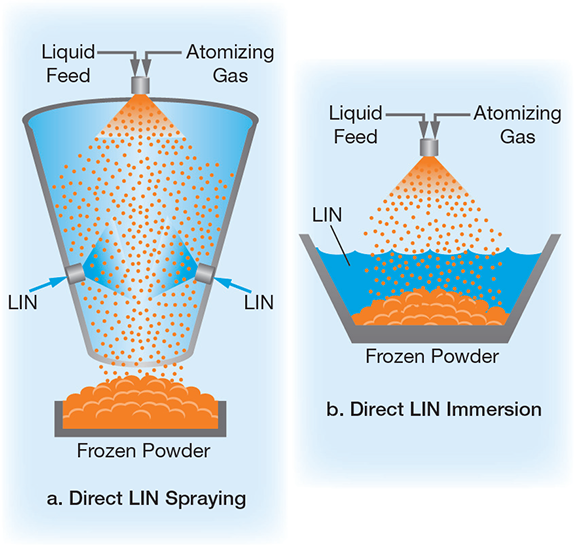
▲Figure 5. In cryogenic spray freeze-drying, atomizing gas creates fine droplets that freeze as they are (a) sprayed with LIN or (b) immersed in LIN.
Direct injection cooling (Figure 7a). LIN is injected directly into the reaction. This method achieves maximum efficiency and is inexpensive to install, but solvent entrainment, foaming, and localized freezing can occur. It is often used in emergencies, because vaporizing LIN can rapidly cool an unsafe or runaway reaction.
Semi-indirect cooling (Figure 7b). LIN flows through either a coil inside the reactor or a reactor cooling jacket. This simple method enables fast cooling and nitrogen recycle. However, drawbacks include lower cooling efficiency and higher costs for cryogenic construction and corrosion-resistant materials.
Indirect cooling with HTF (Figure 7c). In an external heat exchanger, LIN cools a heat-transfer fluid, which then flows through a secondary circuit to cool the reactor. This flexible approach provides accurate temperature control and accommodates large heat loads.
Cryogenic solvent and VOC recovery
Organic solvents and volatile organic compounds (VOCs) can be condensed with LIN and recovered. The ability to recover and reuse evaporated solvents reduces the amount of solvent that must be purchased. The cost savings is typically significant enough for recovery systems to pay for themselves in only a few years. Cryogenic VOC recovery is often a more environmentally friendly and cost-effective way to control VOC emissions than oxidation techniques.
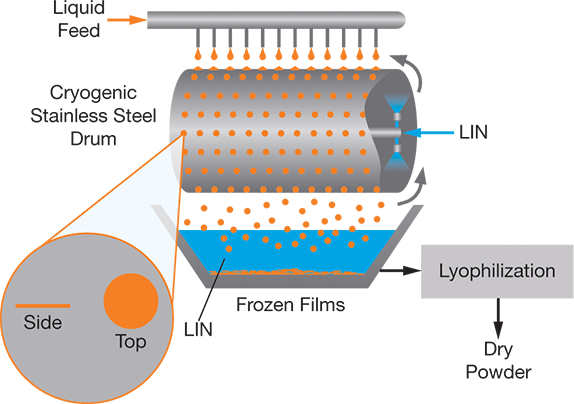
▲Figure 6. In cryogenic thin-film freezing, droplets fall onto a LIN-cooled, rotating, stainless steel drum, where they collapse into flat, thin films as they quickly freeze. Lyophilization dries the frozen films to produce dry powders.
The heart of a cryocondensation system is a heat exchanger, which provides LIN refrigeration, typically through secondary circuit cooling with a heat-transfer fluid or direct LIN injection cooling, to condense solvent vapors or VOCs.
Direct surface cooling, which involves passing LIN through a traditional shell-and-tube heat exchanger, is generally not recommended for VOC recovery operations, especially those driven by environmental regulations. These applications require a higher degree of temperature control and reliability than direct surface cooling can offer. Unstable VOC freezing or heat exchanger plugging can result in a breach in VOC compliance or an unplanned process shutdown, both of which can be serious and expensive incidents.
Secondary circuit cooling with a heat-transfer fluid is a more suitable method, as it allows precise temperature tuning to meet the desired VOC recovery efficiencies. The HTF is loaded into the shell side of the low-temperature condenser, where the temperature is precisely controlled via LIN heat exchange. Concurrently, the VOCs or solvent vapors are condensed inside the condenser by the refrigerated HTF.
Direct LIN injection cooling for VOC recovery is a newer technique that is still under development. LIN is directly injected into the VOC-laden vent streams to freeze the VOCs. A downstream filter collects and separates the frozen VOCs from the vent stream. This technique is especially useful for VOC streams that are subject to strict regulations.
Cryopreservation
Cryopreservation (or cryoconservation) preserves biological samples at cryogenic temperatures for the purpose of prolonged or indefinite storage. The process operates on the principle that low temperatures exponentially reduce the rate of reactions associated with biological activity. Thus, the biological activity of samples at cryogenic temperatures is slowed to the extent that time is, in effect, stopped for the sample. The intention is that the stabilized sample can be warmed to ambient temperature at some point in the future — in some cases, up to 1,000 years later (6) — to resume normal activity.
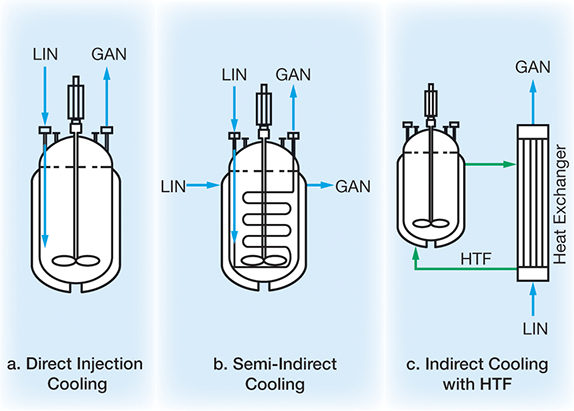
▲Figure 7. LIN can provide refrigeration to highly exothermic or cold-chemistry reactions through (a) direct injection cooling, (b) semi-indirect cooling, or (c) indirect cooling with a heat-transfer fluid (HTF).
Cryopreservation is a growing practice used to store many types of biological materials, including proteins, nucleic acids, cellular structures, single-cell or multicellular organisms, tissues, and organs.
For most samples, the most critical part of the cryopreservation process is the freezing step. If it is too slow, water freezing outside the cell draws out intracellular water molecules through osmotic pressure. The decrease in intracellular water increases the solute concentration inside the cell to potentially lethal levels. Also, slow freezing causes intracellular water molecules to arrange in large crystal structures that can pierce cell membranes and cause irreversible damage to the sample. In general, faster freezing forms smaller ice crystals, which are less damaging to cell structures.
Typically, cryoprotectants (e.g., glycerol, glucose, dimethylsulfoxide) and a cooling rate of 1°C per minute are standard protocols for cryogenic freezing of biological samples (7).
Vitrification is a novel approach for freezing samples for cryopreservation. Cryoprotectants reduce the freezing temperature, and additives increase the viscosity of the sample. The sample is then exposed to an extremely fast freezing regime to vitrify it in amorphous ice and prevent water crystal formation. It is critical that these samples be stored at a cryogenic temperature below the point at which water molecules begin to crystallize.
A general rule of thumb for most cryopreserved samples is to store them below –130°C. The most effective and economical way to store samples is by immersing them in LIN or by cooling them with cold GAN vapor inside a specially designed cryochamber.
Closing thoughts
Since its large-scale commercialization at the turn of the 20th century, nitrogen has become essential to the CPI. Its inerting capability enhances the safety of many operations and the quality of many products. Its use as a cryogen for extremely low-temperature refrigeration continues to grow as cryogenic processes are developed and improved.
The optimization of advanced process controls for liquid nitrogen cooling and freezing systems is an ongoing area of research. Precise control of temperature setpoints (e.g., ±0.5°C) and heat-transfer rates during extremely low-temperature operation (between –150°C and –196°C) is challenging because of heat leakage to the environment, even with vacuum insulation. Efforts are underway to develop more robust control systems capable of delivering extremely low-temperature liquid nitrogen refrigeration in a more precise, yet cost-effective, manner. CEP
LITERATURE CITED
- Carlson, B., et al., “Nitrogen: A Security Blanket for the Chemical Industry,” Chemical Engineering Progress, 107 (11), pp. 50-55 (Nov. 2011).
- Croll, D., and P. Yanisko, “Use Nitrogen Safely,” Chemical Engineering Progress, 108 (3), pp. 44-48 (Mar. 2012).
- Liue, J., and D. Rouse, “Using Liquid Nitrogen to Maximize Lyophilization Manufacturing Capacity,” BioProcess Inter-national, pp. 56-60 (Feb. 2005).
- Ratul, R., and A. A. Baquee, “Pellets and Pelletization Techniques: A Critical Review,” International Research Journal of Pharmacy, 4 (4), pp. 90-95 (Apr. 2013).
- Williams III, R. O., et al., eds., “Formulating Poorly Water Soluble Drugs,” Springer, New York, NY, pp. 486-490 (2012).
- Lopez, E., et al., “Current Frontiers in Cryobiology,” Chapter 19 in “Technologies for Cryopreservation,” InTech, Rijeka, Croatia, pp. 527-540 (2012).
- Thermo Fisher Scientific, Inc., “Thermo Scientific Nalgene and Nuc Cryopreservation Guide,” www.fisher.co.uk/index.php/en/technical-support?view=kb&kbartid=263, pp. 1-12 (Apr. 2011).
ADDITIONAL RESOURCES
Jha, A. R., “Cryogenic Technology and Applications,” Butterworth-Heinemann, Burlington, MA, pp. 10-13 (2006).
Polidoro, J., et al., “Advancing the Art of Spray Drying,” Manufacturing Chemist, pp. 35-37 (June 2012).
Trembley, J., and S. Ivanova, “Liquid Nitrogen Reaction Cooling,” Process Cooling, pp. 11-13 (Oct. 2012).
Boland, T., and B. Nyland, “Practical Low-Temperature Size Reduction,” Powder/Bulk Solids, pp. 32-37 (July 2014).
OSCAR BETETA is an applied research and development engineer in the advanced technology research facility at Air Products (7201 Hamilton Blvd., Allentown, PA 18195-1501; Phone: (610) 481-7576; Email: betetaog@airproducts.com; Website: www.airproducts.com). His focus is on new commercial applications of liquefied gases, particularly liquid nitrogen, in the chemicals, pharmaceuticals, rubber, and plastics industries. In addition, he promotes new technology offerings and provides technical support to customers. He is a member of AIChE and the Tau Beta Pi engineering honor society. Beteta holds a BSE in chemical engineering from Bucknell Univ.
SVETLANA IVANOVA, PhD, is a marketing and applications development manager at Air Products (7201 Hamilton Blvd., Allentown, PA 181951501; Phone: (610) 481-1474; Email: ivanovsm@airproducts.com; Website: www.airproducts.com). She has over 20 years of experience in technology, marketing, and business development. Her technical expertise is in developing new materials and processes for applications such as gas adsorption, water treatment and analysis, and personal care. She is currently responsible for developing and providing industrial gas-related technologies and solutions to the chemical, pharmaceuticals, and biotech industries. Ivanova earned her MS in chemical engineering from Moscow Chemical Technological Univ. and PhD in chemistry from Moscow Institute of General and Inorganic Chemistry of the Russian Academy of Sciences.
Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
