Session Chairs:
- Pelin Hacarlioglu, Chemours
Session Description:
The poster reception will feature a mix of poster presentations from both industry and academia. As attendees mingle between posters, hors d'oeuvres and drinks will also be served.
As process development scientists, we face wide variety of issues from sensitive reactions to complex separations as we move from lab scale research to pilot scale demonstrations. It is essential to create a smooth transition to commercialization to bring a new product to market successfully. This poster session will cover diverse topics from experimental to modeling studies in process optimization to process scale-up issues across industries from pharmaceuticals to specialty chemicals. The purpose of this session is to allow presenters and symposium participants to have lively discussions on specific topics as well as give them immense opportunity to learn from each other’s experiences.
Schedule:
The Poster Session will formally take place from 5:00pm - 6:00pm. Posters will be available for viewing throughout the full reception.
Abstracts:
Design and Commissioning of a Vapor Phase Miniplant System with Laboratory Fume Hood Constraint
Michael Molnar, Dow-Corning
Exothermic reactions involving vapor reagents under pressure are common within chemical manufacturing, but scale-up of these systems usually goes through multiple stages of development (lab → miniplant and/or pilot plant → commercial). Safe management of exotherms and undesired consequences of the chemistry such as side reactions must be well understood to allow for a robust process implementation and safe process operation.
Manufacture of some silanes involve exothermic chemistries yet kinetic limitations often force operating temperatures exceeding 300 °C, which pushes the upper boundary of conventional heat transfer fluids if used to dual purpose heat and manage the exotherm [1, 2]. To accomplish a process evaluation at miniplant scale, an OEM-supplied, resistance-heated, fluidized sand bath system was modified to accommodate a multi-tubular reactor arrangement while operating at temperatures up to 600 °C and pressures up to 150 psig. This arrangement allowed for very high external heat transfer coefficients which supported exotherm mitigation, even in the case where the tubes operated as fixed beds. Flow was oriented single pass to avoid extended post-reaction dwell time and extended preheat for the chemistries of interest, but this made mechanical implementation within the sand bath more challenging. Process feedback on the bed temperature profile was accomplished with multiple axial multi-element thermocouples, each housed within a specialized thermowell. Design considerations were placed on removing the catalyst materials following the reaction campaign while allowing for adjustable clearance in the position of the sand bath through use of a specialized lift cart. This poster highlights the challenges associated with both designing and commissioning the system within the space constraints of a laboratory hood.
[1] Koerner, G., “Introductory Remarks on the Peculiarities of Silicone Chemistry” in Silicones Chemistry and Technology, Bayer AG, Th. Goldschmidt AG, Wacker-Chemie GmbH, and Haus der Technik, editors, CRC Press, 1991.
[2] Feldner, K., “Chemistry and Technology of Direct Synthesis” in Silicones Chemistry and Technology, Bayer AG, Th. Goldschmidt AG, Wacker-Chemie GmbH, and Haus der Technik, editors, CRC Press, 1991
Scale up of PEEK Hollow Fiber Membrane Contractor for Post Combustion CO2 Capture
Shiguang Li, Travis Pyrzynski, Naomi B. Klinghoffer, James L. Aderhold, Howard S. Meyer, Yong Ding, and Benjamin Bikson, Gas Technology Institute
Poly(ether ether ketone) (PEEK) hollow fiber membrane modules were scaled up from 2 inch diameterand 16 inch long to 4 inch diameter and 58 inch long (a factor of 90 increase in membrane area) for CO2 capture from flue gases using membrane contactor process, which combines advantageous features of both absorption and membrane processes. Field test of a 4-inch-diamter module with activated methyldiethanolamine (aMDEA) solvent demonstrated greater than 90% CO2 removal. The mass transfer coefficient in the absorption step was 1.2 (sec)-1, which is over an order of magnitude greater than that of conventional column contactors. The membrane module was further scaled up from 4 inch to 8 inch diameter (a factor 3 increase in membrane area). Single-gas permeation measurements indicated that 8-inch modules have an intrinsic CO2 permeance as high as 2,150-2,670 GPU. One 8-inch-diameter module was tested in membrane contactor mode for CO2 capture using a simulated flue gas. Carbon dioxide removal rate of 91.3% was achieved with a mass transfer coefficient of 1.5 (sec)-1. Parametric tests indicated CO2 flux and capture rate increased with increasing feed pressure, solvent flow velocity and solvent temperature. PEEK hollow fiber membrance contactor is effective in capturing CO2 from low CO2-concentration feeds, showing its high potential for natural gas flue gas CO2 capture.
Enabling Rapid Process Development for the Endgame Steps of an API
Anne Mohan, Azzeddine Lekhal, Russell Hensley, David Lamberto, Ivan Lee, Shane Grosser, Alexei Kalinin, Jingjun Yin, Tetsuji Itoh, Bangping Xiang, Margaret Figus, Yu He, Liam Corcoran, Eamonn Joyce, Thorsten Konekamp, Mick Lucey, Nick Rogus, Ruth Rosenthal, Khateeta Emerson, Glenn Spencer, Laura Artino, and Rich Varsolona, Merck & Co.
The 2 endgame steps of an API synthesis involves a double Suzuki coupling reaction, the product of which is isolated by a mandelate salt formation, followed by a salt break and precipitation of the neutral API. The Suzuki reaction of the penultimate step is particularly challenging both to execute and to understand due to the oxygen sensitivity of the catalyst and starting material, and to the sheer amount of impurities which can be formed during the reaction, respectively. The high number of impurities formed during the reaction makes the mandelate salt formation used to isolate the Suzuki product equally challenging, since impurity rejection is critical. In parallel with the process development for the Suzuki, the Pure step has faced several fluctuations over the last 2 years. The changing landscape of the product market has significantly impacted the Pure step specifically in the last ~6 months due to variations in both API demand and drug product formulation. The potential shift from spray drying to conventional formulation has significantly impacted the process development and desired physical attributes of the API. This poster will detail the comprehensive and hefty process development work which was executed to support the endgame steps, and their respective impacts on the broader program.
Concepts for Scale-up in a Loop Reactor
Aaron Shinkle, The Dow Chemical Company
Batch chemical operations are regularly used for fine and specialty chemicals due to versatility of the reactor for use on the next product. However, continuous processes have begun to gain traction. One particular kind of continuous reactor is the loop reactor which is a vessel-less CSTR involving a loop of piping which the process fluid is recirculated around at a much faster rate than product is withdrawn. Loop reactors are commonly considered when one of the following conditions are met: hazardous chemicals are used, significant heat removal is needed, reaction rate is fast, significant decomposition reactions occur, or high local mixing is required. This poster discusses several design concepts and key challenges that may be useful when setting up lab-scale loop reactor systems. Specific focus is placed on the role of multiple phases in the loop reactor. Examples of lab-scale loop reactors built at Dow Chemical are given along with statistics showing how the loop reactor process compares with the equivalent batch process.
Development of a Robust, Scalable Process for the Manufacturing of Organogels
Justin Walker and Mahesh Sawant, The Dow Chemical Company
The manufacturing of organogels requires precise control of process parameters such as dispersion, agitation, heat transfer, and packaging in order to achieve a gel product with consistent/repeatable properties required to impart desirable features (e.g., soft, smooth, spreadable, etc.) to the formulations of our customers. Deviations from ideal processing conditions may result in the formation of gels which are hard, lumpy, tacky, oily, or otherwise unacceptable. In addition, cGMP or other regulatory requirements often place constraints on the process equipment selection (materials of construction, cleanability, environmental controls, etc) for the production of such gels, frequently necessitating the employment of different unit operation designs that are volume or scale dependent, requiring careful analysis and modification of the process at each stage of development from laboratory scale through pilot scale to full scale production to ensure success.
In order to support the effective development of this class of products, Dow’s unique collaborative engine of Research and Development, business, and manufacturing expertise brings the processing and product capabilities and know-how needed to develop robust scalable process designs to facilitate the economical production of organogels that provide the innovative solutions our customers demand.
Utilizing a combination of Computational Fluid Dynamics (CFD), laboratory experiments, and pilot plant trials, the Dow team have developed and effectively implemented process technology which has facilitated the successful advancement of such organogel materials to full scale production.
Innovative Process Development for Next Generation Fluorochemical Products
Michael Bradley, Chemours
As a global leader in Flouorochemicals, Chemours is continuously evolving its chemistry to provide more environmentally sustainable and lower global warming potential (GWP) products. Utilizing pilot scale studies, analytical techniques, physical property measurement, and steady state process modeling, we are able to develop timely and innovative solutions aimed at meeting the market demand while maximizing plant yields and minimizing capital investment requirements. Examples of the use of these tools in the development of the latest generation of product offerings, hydrofluooroolefins (HFOs), are highlighted in this poster.
Commercializing a Low Global Warming Potential Refrigerant
Karl Krause, Chemours
Regulations enacted in Europe in the mid-2000’s requiring a low global warming potential (GWP) refrigerant in mobile air conditioning applications had industry racing to develop a replacement for the incumbent HFC-134a. This presented a challenge to not only identify, test, and qualify a next generation refrigerant, but also to develop and commercialize processes to manufacture it under a very aggressive schedule. Using innovative product and process development, Chemours successfully introduced Opteon® yf within 7 years of the directive and was the first bulk producer of HFO-1234yf which has a GWP of less than 1. This poster describes the journey from initial candidate evaluation to market acceptance to commercialization of Opteon® yf focusing on the process development for world’s the first large scale bulk manufacturing facility. Further innovations to meet increasing demand as use of HFO-1234yf around the world ramps up will also be presented.
Sizing Control Valve for Lab Scale Laminar Flow
Grant Girouard and Paul Martin, Zeton, Inc.
Control valves for low flows of liquids, even of low viscosity, must be sized to take into account laminar or transitional flow in the valve trim.
We have found that the equation below allows the accurate sizing of control valves with seat-guided type trims for laminar, transitional and turbulent flow:
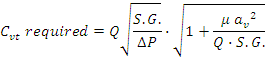
A single new parameter  is used to adjust the sizing of families of valve trims of similar geometry, for viscosity dependence in the laminar and transitional regions.
is used to adjust the sizing of families of valve trims of similar geometry, for viscosity dependence in the laminar and transitional regions.
This new parameter has a value of approximately 0.14 for seat-guided annular needle-type trims such as those used in Badger Research Control Valves and for Jordan LowFlow control valves for trims of Cv. 0.06 and smaller. A single Samson valve of similar trim type was tested and found to have an  of 0.18, which we suggest to use for valves of similar design for conservative trim sizing in the absence of other data. We found
of 0.18, which we suggest to use for valves of similar design for conservative trim sizing in the absence of other data. We found  to be approximately constant with varying stem position for trims of this geometry.
to be approximately constant with varying stem position for trims of this geometry.
For seat-guided vee-groove control valve trims manufactured by Emerson (Baumann), the value of  is approximately 0.06 at full open, and tends to smaller values as the valve is closed.
is approximately 0.06 at full open, and tends to smaller values as the valve is closed.
This equation can be solved to be explicit in Q and tends to the normal turbulent flow Cv sizing equation when the product 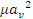 is sufficiently small.
is sufficiently small.
Using Reaction Calorimetry for Safe Process Scale up
Maria Birau, Xerox Research Centre of Canada
Sodium borohydride (NaBH4) is a versatile reagent used to convert carbonyl groups into alcohols and it can be used in aqueous or alcoholic solutions. It is also known that sodium borohydride reduction is an exothermal reaction with vigorous hydrogen evolution and this can pose a significant problem for scaling-up of bench experiments. Calorimetric information is very useful in helping to identify issues related to heat transfer, fouling, precipitation, viscosity changes. Therefore a hydrogenation study was performed in an OptiMaxTM HFCal instrument using a model carbonyl compound and sodium borohydride in: (a) solid form (powder) and (b) liquid form (12 wt.% aq. solution stabilized with 14 M NaOH). The findings from this study were applied to the safe scale-up of hydrogenation reaction of tetraformyl dimethyl tetraphenylbiphenyl diamine to tetrahydroxy dimethyl tetraphenylbiphenyl diamine.
Forward Osmosis: Energy Efficient Water Recovery from Industrial Effluents
George Kolliopoulos, University of Toronto
To date, in Ontario hazardous industrial effluents are shipped to Ohio in the US at a significant cost. The amount of such streams generated every year is approximately 2 million m3. Recovering water from hazardous effluent streams with a low-cost process would enable clean water recycling in industrial operations and reduction of the volume of hazardous effluents requiring disposal or further treatment. Forward Osmosis (FO) is an innovative membrane process, able to recover water from industrial effluent solutions containing up to 240,000 ppm total dissolved solids (TDS) at low cost. An engineered concentrated draw solution (CDS) selectively draws water molecules from an effluent solution, across the pores of a semi-permeable membrane through a spontaneous osmotic phenomenon. Water flows without any external energy input, due to the osmotic pressure gradient generated between the two solutions. Eventually, the CDS becomes diluted and must be treated to separate the draw solutes from the water recovered. This step requires energy input. However, if the CDS consisted of a thermolytic salt, such as trimethylamine (TMA) bicarbonate, which decomposes to gaseous TMA and CO2 upon moderate heating, the energy input would be small. In the present work, the effectiveness of an aqueous carbonated trimethylamine solution as the CDS in FO is evaluated. Its ability to draw water from industrial effluents as well as the separation efficiency of the draw solutes from the water recovered are also reported. Finally, the overall energy requirements in a continuous integrated FO process are estimated, using results from a micro pilot unit and the OLI-Flowsheet software.
Catalyst Development Efforts during Scale-up of the THF ex-Butane Process
Greg Sisler, DuPont
In the 1990's, DuPont embarked on an program to develop a lower-cost process for tetrahydrofuran (THF). A 2-step process based on butane was selected for scale-up. It was believed this process could provide lower cost THF than the existing Reppe process which is based on natural gas through formaldehyde and acetylene. In step 1 of the butane-based process, butane was partially oxidized to maleic anhydride in a circulating fluidized bed reactor using attrition resistant vanadium phosphorus oxide (VPO) catalyst. The maleic anhydride was scrubbed and recovered as maleic acid. In step 2, the maleic acid was hydrogenated to THF on a supported precious-metal catalyst. The overall process and the catalyst manufacturing for each process step were scaled up from lab to pilot to commercial scale. This poster will focus on selected challenges and successes around scale-up and performance of the catalyst.
Implementation of a Commercial Scale Filtration using Lab Data
David Couling, The Dow Chemical Company
Filtrations, although critical to many fine chemical processes, are often approached with some trepidation during initial scale up due to the significant challenge of developing lab-scale data that translates flawlessly to scale. Some primary factors that contribute to the inaccuracies include cake thicknesses that may vary in depth, particle morphologies that may be altered due to scale-related changes in agitation or heat transfer, and the filtration equipment itself that may be different from what was demonstrable in the lab. As a result, the filtration flux is difficult to predict, many times leading to undesirable results. Here, a recent implementation is presented in which a combination of lab experiments and theory led to the successful scale-up of a filtration from a 1.5-inch pocket filter to a 4-foot centrifuge.
Crystallization in Process Chemistry: Applying Simple PAT Tools
Terry Redman, Mettler-Toledo
Crystallization is a unit operation offering a method to isolate and purify products in a single step. At the same time, crystallization is a complex process where it is possible to have multiple process variables interacting to affect the properties of the size, shape, yield, form and purity of the crystal product.
Many published studies have shown the value of process analytical technologies (PAT) for drastic improvement in the ability to characterize, optimize, and scale-up, and control robust crystallization processes. However, many process analytical techniques, such as FBRM and ATR-FTIR spectroscopy, provide complex data that sometimes limits their adoption to crystallization experts and specialized engineering groups.
Real-time microscopy with simple image analysis has been developed to provide a direct measurement of the relative backscatter index (RBI), offering an opportunity for every scientist to utilize Process Analytical Technology PAT in the design, analysis, optimization, and control of crystallization processes.

