Tissues and organs inside our bodies are incredibly complex at the cell level. Cells can be differentiated by type and functional state. This biological insight has combined with technological advancements to help usher in the era of single-cell diagnostics for tissue-based diseases. Single-cell approaches enable researchers to rapidly and comprehensively analyze cells for type and function, which cannot be done for intact tissues.
However, a major challenge of this approach has been the need to dissociate the tissue into single cells. Current procedures are labor- and time-intensive, requiring mincing of tissues into small pieces with a scalpel, a long enzymatic digestion step, crude mechanical treatments, and filtering. Extraction using the traditional approach is inefficient and yields poor cell quality.
Technological advances to aid this multistep workflow have been minimal, targeting only one step. A new technology that could perform the entire process in a rapid, gentle, thorough, and automated manner would dramatically advance single-cell diagnostics and boost their clinical potential, thus paving the way for powerful new personalized treatments. For example, this technology could provide valuable insight into cancerous and non-cancerous cells that could help scientists predict disease progression and select the most effective therapies to bring about a cure for cancer. Tissue engineering and regenerative medicine would also benefit from improving procurement of healthy and functional primary, progenitor, and stem cells from various organs and tissues to serve in tissue constructs and cell-based therapies.
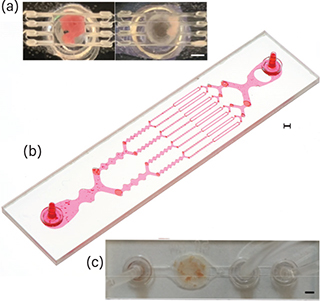
▲ Researchers have created microfluidic devices for (a) digestion, (b) disaggregation, and (c) filtration of tissue samples to generate single cells. Mouse kidney samples are represented in each photo and red dye was added to the disaggregation device. Scale bars are 2 mm for each image.
To meet this challenge, Jered Haun’s group at the Univ. of California, Irvine, has developed a novel microscale fluidic device that can perform the entire tissue dissociation workflow while gently and efficiently producing single cells.
Microfluidic chips operate at physical length scales comparable to biological constituents (e.g., cells and molecules), which is typically tens of microns. However, tissue samples start at the millimeter scale, at least. This has led Haun’s group to develop a series of devices that can bridge the length scale gap.
The first device facilitates enzymatic digestion of specimens using fluid jets that cut the tissue down into smaller fragments. Then, a second device generates shear stresses to break tissue fragments into smaller units — from cell aggregates to clusters, and then finally to single cells — using an array of branching channels that repeatedly expand and constrict. The third device uses nylon mesh filters to eliminate any remaining aggregates using both traditional filtration and a novel dissociation mechanism.
Each device can improve tissue dissociation procedures on its own. However, all three devices are currently being combined into an integrated platform, the first of its kind, that can perform the entire tissue-to-cell workflow. Early tests have shown that the integrated device platform can extract more than 20,000 single, viable cells per milligram of tissue, which is two-fold to ten-fold greater than traditional methods. Alternatively, the device can produce similar numbers to traditional methods in one-fourth the time (i.e., 15 minutes). Recent work has validated the platform using an advanced analysis method, single-cell RNA sequencing.
This work is being performed by the Center for Advanced Design and Manufacturing of Integrated Micro-fluidics (CADMIM). This collaborative center consists of two university sites (Univ. of California, Irvine and the Univ. of Illinois at Chicago) and 11 corporate members that develop microfluidic platforms for use in assessment of the environment, food and water supplies, and ultimately human health and safety.
Each microfluidic device is fabricated from multiple layers of hard plastic using a commercially scalable laminate process provided by CADMIM member ALine, Inc. Using this process has helped the team produce commercial-quality devices at a sufficiently low price point that the device is disposable, which is needed for clinical applications.
The tissue dissociation devices are being commercialized through Kino Discovery, a startup company.
This research is funded through the NSF’s Industry-University Cooperative Research Centers (IUCRC) Program.
This article was prepared by the National Science Foundation in partnership with CEP.

Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
