Biomanufacturing, particularly to produce biologic pharmaceuticals, has entered an era of intensified processing in which mammalian cells are cultivated to very high cell densities and with very high product titers (i.e., higher drug concentrations in the cell-free culture broth). This allows the use of smaller, cheaper bioreactors and lowers purification costs.
A critical requirement for keeping cells at these high densities and producing high product levels is having high nutrient concentrations in media and in feed streams, particularly amino acids, salts, and sugars. Often, the optimal media for a particular cell culture has concentrations of some species (usually amino acids) that are close to or above their pure component solubilities in water. Because media formulations often have twenty (or even a hundred) components, knowing the solubilities of all the components in these complex mixtures can be a problem. Without this understanding, formulating stable multi-component media is difficult, posing a significant challenge to process design and scale-up. Media formulations have relied on extensive trial-and-error experiments that cause bottlenecks in biomanufacturing process development.
A phase-equilibrium model that can predict solubilities in such systems could provide an efficient path to media formulation. Modeling these multi-component solutions requires knowledge of all the different intermolecular interactions and their impacts on solubility. For media, these interactions include (at a minimum) dispersion forces, ionic forces, and the effects of the formation of strong chemical complexes. For example, a solution of one amino acid (AA) with one salt (XY) in water can have nine or more different species present in solution, including the neutral AA, the AA zwitterion, AA+ or AA– (depending on the pH), molecular XY, X+, Y–, as well as AA+Y– and AA–X+ (the salts of the amino acid with the ions of XY). A typical medium with twenty components will have 50 to 100 different species in solution due to the formation of these chemical associations.
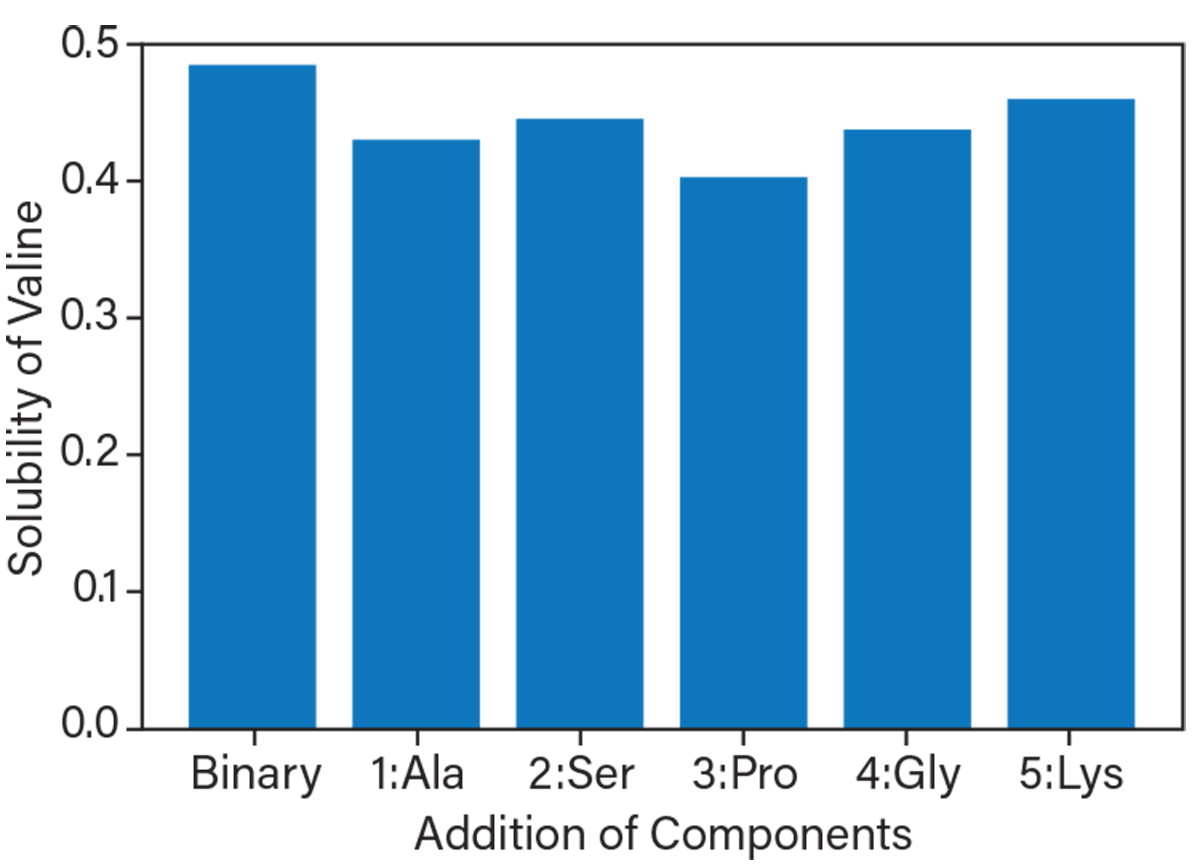
▲ The ability to predict the behavior of complex multi-component solutions will vastly simplify media formulation in process development and scale-up. This figure shows the change in solubility of the amino acid valine as other amino acids are added sequentially. The bar for Binary is the solubility of valine in water. The next bar labeled 1:Ala shows that adding 1 M alanine decreases the valine solubility. The bar labeled 2:Ser shows how the addition of 1 M serene affects the solubility of valine in a solution that also contains valine and alanine. The same applies to the last three bars for 1 M proline, 1 M glycine, and 0.5 M lysine.
The importance of this problem to the success of high-titer mammalian cell cultures has been recognized by the Advanced Mammalian Biomanufacturing Innovation Center (AMBIC), an Industry-University Cooperative Research Center (IUCRC) funded by the U.S. National Science Foundation (NSF). AMBIC is also supported by research funding from 33 biopharmaceutical companies, and it sponsors fundamental, pre-competitive research at five different universities. AMBIC research has developed parameter databases, a thermodynamic model, and media formulation tools that promise to significantly simplify and accelerate process design and scale-up.
Current efforts are focused on expanding the database of all the molecular parameters needed to predict speciation and solubilities in solution. This database currently has nearly a thousand different possible media constituents. The Center’s thermodynamic model (available as a user-friendly software package) can predict speciation of hundreds of components into various complexes with metal ions. It also can be used to obtain actual nutrient availability for concentration-dependent transport into cells by predicting the percentage of each nutrient being tied up in complexes. The thermodynamic predictions have been validated experimentally by some AMBIC member companies with >85% prediction accuracy.
AMBIC media formulation tools are able to predict the optimal concentrations, storage temperatures, and pH of media and media concentrates. They also offer algorithms to predict the optimal order-of-addition of all the different components of the desired media formulation such that no precipitation occurs during preparation or storage of the media or media concentrates.
“The algorithms, databases, and software developed for predicting media component solubility have been a major success for the AMBIC industrial community,” says Matt Zustiak, the Director of Cell Culture Development at Thermo Fisher Scientific and Chair of the AMBIC Industrial Advisory Board. “The models have been adopted by several member companies, and as the industry continues to intensify processes to increase biologics manufacturing output, this knowledge will undoubtedly help to accelerate those efforts.”
This research was supported by the Industry-University Cooperative Research Center (IUCRC) program of the NSF.
This article was prepared by the National Science Foundation in partnership with CEP.

Copyright Permissions
Would you like to reuse content from CEP Magazine? It’s easy to request permission to reuse content. Simply click here to connect instantly to licensing services, where you can choose from a list of options regarding how you would like to reuse the desired content and complete the transaction.
