(555a) Solubility Measurements of Dipeptides at Different Temperature and Solution Environment: An Investigation on Peptides Sequence Effect on Solubilities
AIChE Annual Meeting
2023
2023 AIChE Annual Meeting
Engineering Sciences and Fundamentals
Thermophysical Properties and Phase Behavior II
Wednesday, November 8, 2023 - 3:30pm to 3:47pm
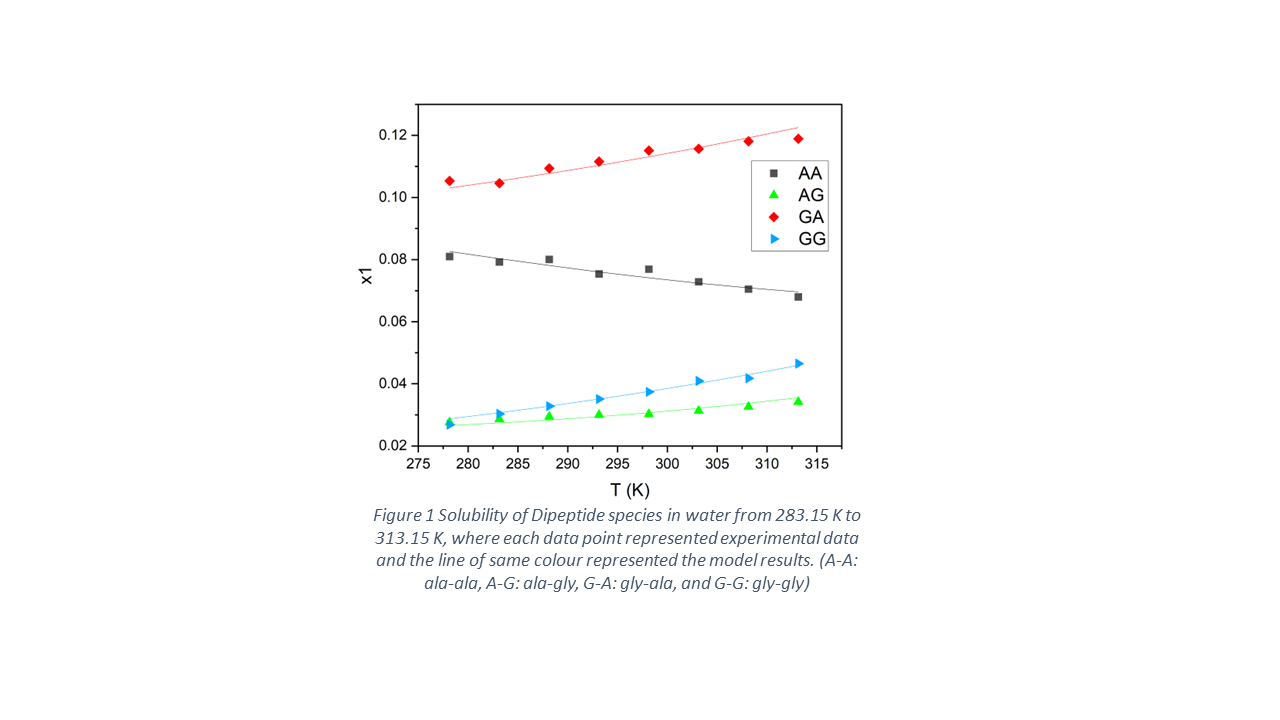
The most common crystallisation methods are cooling crystallisation and antisolvent crystallisation. The former uses temperature change to get the solids out and the latter relies on addition of antisolvent. In this work, the solubility of four dipeptides (ala-ala, ala-gly, gly-ala and gly-gly) are measured in water at in the temperature range from 278.15 K to 313.15K, and at various antisolvent (ethanol and DMSO) concentration.
The solubility order of the 4 dipeptides in water is: gly-ala > ala-ala> gly-gly > ala-gly, which is in accordance with literature data reported by Do, Franke et al. 4 Molecular dynamic simulation was done for the calculation of solvation free energy to explain the difference between species. The solvation free energy order also conformed the solubility order: gly-ala < ala-ala< gly-gly < ala-gly, with lower solvation free energy indicating higher solubility.
It was found that alanine group in the peptide chain not only affected the solubility value of the species, but also the temperature dependence of solubility. For gly-gly, its solubility in water increases significantly as temperature increases from 278.15 K to 313.15 K. While when there was alanine component in the peptides chain, the solubility curve became flatter and temperature effect was reduced. As for ala-ala, its solubility decreased as temperature rose. The enthalpy change of dissolution in water was measured by TAM calorimeter, which was -20 kJ/mol. This result can relate the unusual solubility behaviour to le chatelier's principle.
The solubilities of dipeptides in antisolvent solutions of concentration range from 0 to 80 % by weight were also reported at 298.15 K. DMSO and ethanol were showing similar effect on the solubility reduction. Overall, gly-gly’s solubility was affected the most by the addition of antisolvent, where only less than 5% species stayed in the liquid state when 50% antisolvent was added. Gly-ala was least affected, but solubility was still reduced to less than 10% at 50% antisolvent concentration.
The solubility date was fitted to non-random two-liquid (NRTL) model parameters, to predict the phase equilibrium conditions of the mixture at different temperatures. The NRTL model was proved valid for aqueous and ionic systems5. The temperature dependent interaction parameters between water and dipeptide molecules were obtained from solubility data of dipeptides in water at different temperature. As for the interaction parameters between water and antisolvent/antisolvent and dipeptide, those were optimised into temperature independent form.
In summary, the solubility of 4 dipeptides based on alanine and glycine were studied at different temperature and antisolvent concentration, with NRTL model fitted to extend the solubility profiles at various temperature and solution environments. This research establishes a thermodynamic foundation for the further design of peptides crystallisation process, providing a flatform to explore more the relationship between peptide sequences and their physicochemical properties.
1 Patrick Vlieghe, V. L., Jean Martinez, and Michel Khrestchatisky. Synthetic therapeutic peptides: science and market. Drug Discovery Today 15 (2010).
2 Henninot, A., Collins, J. C. & Nuss, J. M. The Current State of Peptide Drug Discovery: Back to the Future? J Med Chem 61, 1382-1414 (2018). https://doi.org:10.1021/acs.jmedchem.7b00318
3 Agyei, D., Ongkudon, C. M., Wei, C. Y., Chan, A. S. & Danquah, M. K. Bioprocess challenges to the isolation and purification of bioactive peptides. Food and Bioproducts Processing 98, 244-256 (2016). https://doi.org:10.1016/j.fbp.2016.02.003
4 Do, H. T. et al. Measurement and modelling solubility of amino acids and peptides in aqueous 2-propanol solutions. Phys Chem Chem Phys 23, 10852-10863 (2021). https://doi.org:10.1039/d1cp00005e
5 Carr, A. G. & Tester, J. W. Prediction of the solubility of quartz in salt solutions from 25°C to 900°C using the 3-parameter Non-Random Two-Liquid (NRTL) model. Fluid Phase Equilibria 337, 288-297 (2013). https://doi.org:https://doi.org/10.1016/j.fluid.2012.10.009