(471e) Using Machine Learning to Decompose Large-Scale MILP Supply Chain Models
AIChE Annual Meeting
2023
2023 AIChE Annual Meeting
Computing and Systems Technology Division
Planning, Scheduling, Supply Chain and Logistics II
Monday, November 6, 2023 - 1:54pm to 2:15pm
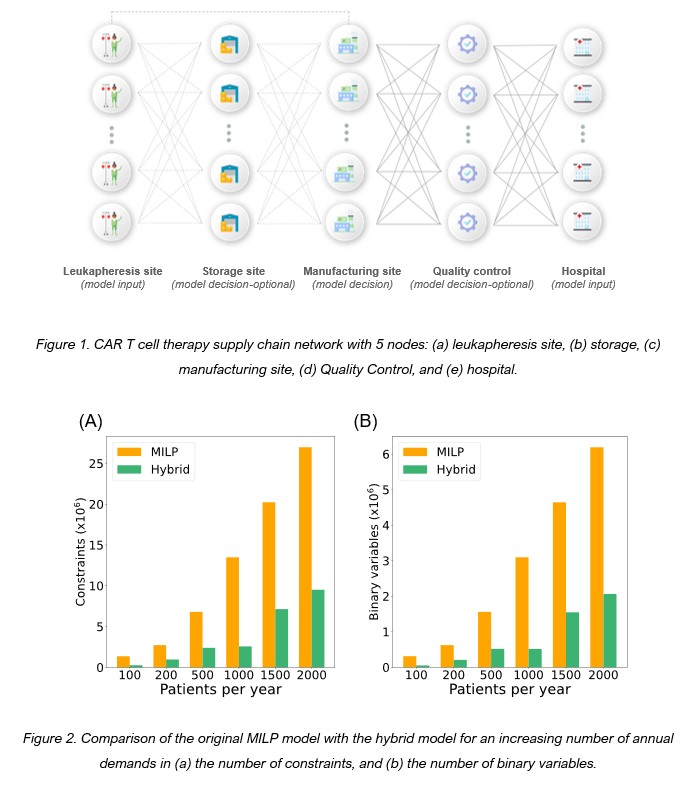
In this work, we present a hybrid model that describes the CAR T-cell supply chain based on Mixed Integer Linear Programming (MILP) and Artificial Neural Networks (ANNs). We utilize our previously developed MILP formulation that describes the CAR T-cell therapy supply chain and tracks each patient/sample/therapy throughout the CAR T-cell lifecycle [3]. The model is used for the identification of optimal supply chain network structures (planning), and the optimal coordination of therapies in the manufacturing facilities (scheduling), given randomized demand profiles of patients being treated at clinical centers in the UK throughout the year. From a modeling perspective, the patient-specific nature of these therapeutics leads to complex large-scale models that grow exponentially as the demand increases. Indicatively, for demands of 2000 therapies per year, the model consists of 26,975,374 linear constraints and 6,190,134 binary variables (Fig. 2). We, therefore, harness the potential of machine learning to decrease the computational complexity of the MILP model. The data-driven part of the model is responsible for strategic planning by forecasting the optimal supply chain structure [4–5] based on uncertain annual patient demand profiles. The MILP model becomes then a subproblem of the original MILP, as it is solved for a fixed supply chain network determined by the ANN and it is now solely responsible for the detailed scheduling of the patient samples to the predefined manufacturing facilities.
We assess and evaluate the performance of different artificial neural network (ANN) configurations including feed-forward neural networks and convolutional neural networks and train them to perform multi-label classification, where the labels are the candidate manufacturing facilities and the possibility of MILP infeasible solutions due to limited facility capacity. To ensure that the hybrid model is transferable, we train the ANNs using different probability distributions for the demand profiles to account for the demand uncertainty in the emerging cell and gene therapy sector. The results showcase a reduction of up to 81% and 83% in the number of linear constraints and binary variables, respectively, for examined (Fig. 2).
Keywords: supply chain optimization, MILP decomposition, machine learning
Acknowledgements
Funding from the UK Engineering & Physical Sciences Research Council (EPSRC) for the Future Targeted Healthcare Manufacturing Hub hosted at University College London with UK university partners is gratefully acknowledged (Grant Reference: EP/P006485/1). Financial and in-kind support from the consortium of industrial users and sector organizations is also acknowledged.
References
[1] C. M. Young, C. Quinn, & M. R. Trusheim, 2022, Durable cell and gene therapy potential patient and financial impact: US projections of product approvals, patients treated, and product revenues, Drug Discovery Today, 27(1), 17–30.
[2] M. M. Papathanasiou, C. Stamatis, M. Lakelin, S. Farid, N. Titchener-Hooker, N. Shah, 2020, Autologous CAR T-cell therapies supply chain: challenges and opportunities?, Cancer Gene Ther., 27, 799-809.
[3] N. Triantafyllou, A. Bernardi, M. Lakelin, N. Shah, M. M. Papathanasiou, 2022, A digital platform for the design of patient-centric supply chains, Scientific Reports, 12, 17365.
[4] D. Goettsch, K. K. Castillo-Villar, M. Aranguren, 2020, Machine-Learning Methods to Select Potential Depot Locations for the Supply Chain of Biomass Co-Firing, Energies, 13, 6554.
[5] B. Abbasi, T. Babaei, Z. Hosseinifard, K. Smith-Miles, M. Dehghani, 2020, Predicting solutions of large-scale optimization problems via machine learning: A case study in blood supply chain management, Computers & Operations Research, 119, 104941.