(592g) Performance Evaluation of Membrane Module for CO2 Separation from Ambient Air
AIChE Annual Meeting
2022
2022 Annual Meeting
Environmental Division
Design and Analysis of Carbon Capture and Negative Emissions Technologies - Experimental
Thursday, November 17, 2022 - 10:06am to 10:27am
The entire mechanism of the gas transport from upstream to downstream of polymeric membrane consists of three steps (i) adsorption on the upstream boundary (ii) diffusion through membrane matrix and (iii) evaporation from the downstream boundary. The mechanism of transport of gas molecules through the membrane depends on the structure and material of the membrane. The overall module performance is dependent on operating conditions (such as pressure difference), selectivity of gases, and membrane area and thickness.
In this study, carbon dioxide (400 ppm) is separated from a binary mixture with nitrogen to reduce the amount to 300 ppm which is considered as the concentration of CO2 in the air during the pre-industrial era. As a result, the concentration of CO2 in the permeate stream is enhanced to many folds which can be utilized for other purposes. Separation performance is evaluated based on the pressure ratio between the upstream and downstream, membrane parameters (area and thickness), and the permeability of components or selectivity. The membrane thickness of 1 µm is used. The pressure ratio of 55 (upstream pressure: 110 kPa, downstream pressure: 2 kPa) is kept constant for all simulations. The single-stage separation process with the co-current flow and constant permeability flux model is simulated to assess the sensitivity of selectivity which is the ratio of the permeance of two components.
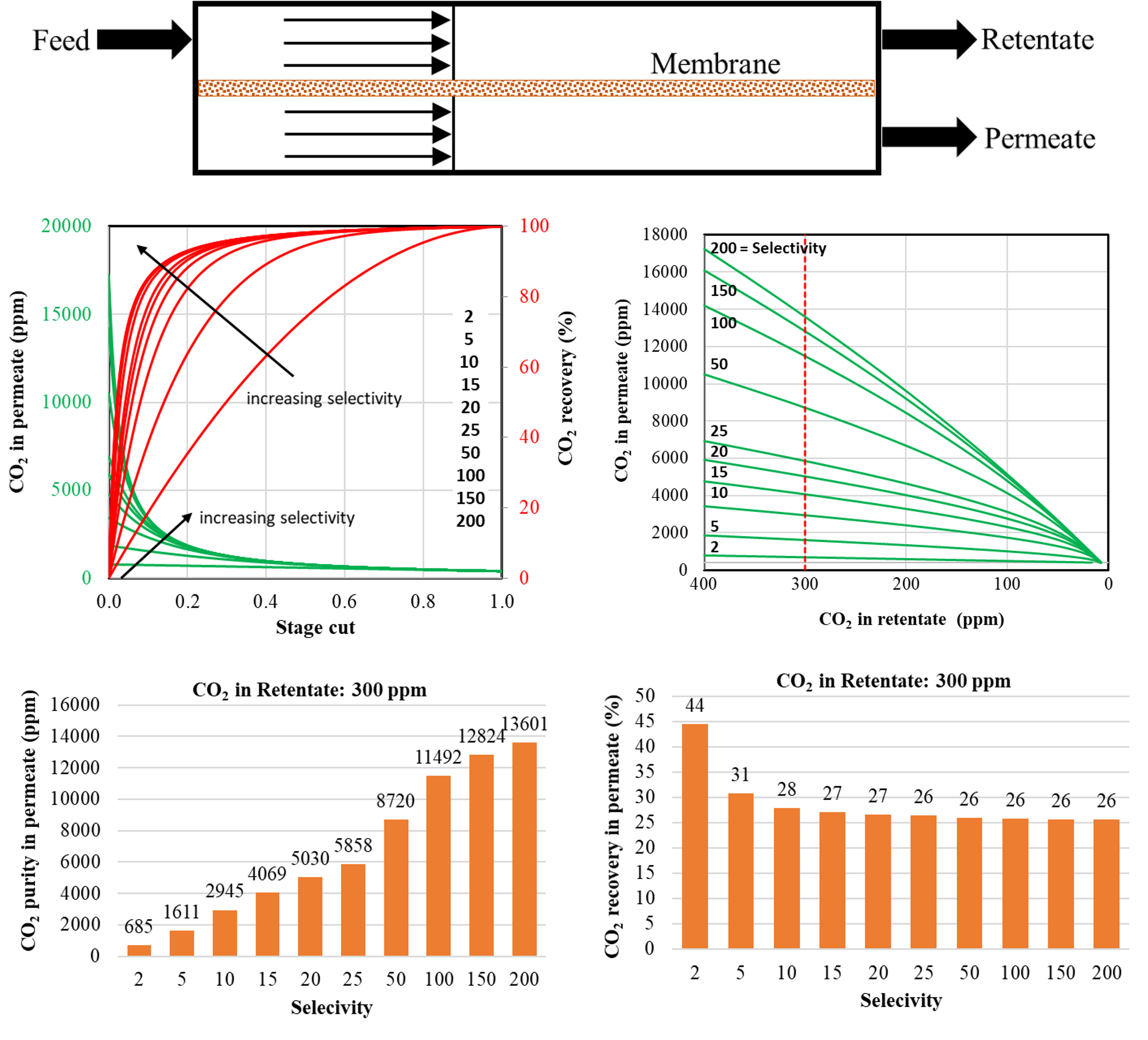
The flow rates and concentrations of all components in the retentate and permeate are obtained from the simulations for different conditions. The separation is influenced by the membrane area when other parameters are fixed. It is often practical to use stage cut which is defined as the ratio of total permeate flow rate and feed flow rate instead of membrane area to compare different simulation results on the same scale. It is noted that the stage cut varies between 0 to 1 with the membrane area. The purity of CO2 is also analyzed which is defined as the concentration of CO2 in permeate. Another parameter is the amount of CO2 in permeate which is quantified by the recovery factor (ratio of CO2 in permeate and CO2 in feed).
In this co-current flow scheme, the retentate stream is in equilibrium with permeate stream on the other side of the membrane. The CO2 concentration in the retentate and permeate decreases along the membrane. However, the total flow rate also increases thus increasing the stage cut as a result and enhancing the CO2 recovery factor. This is a trade-off situation between the purity and recovery factor of CO2 in the permeate stream. This can be fixed by setting the CO2 concentration in the retentate to a certain value such as 300 ppm. Therefore, the membrane area is designed to achieve a specific amount of separation. For a fixed amount of separation, the higher CO2 concentration in the permeate is achieved for higher selectivity. For example, CO2 concentration in the permeate is 685 ppm for selectivity of 2 and 13601 ppm for selectivity of 200. The CO2 recovery factor doesn’t change much for selectivity above 20. It is 44% for selectivity of 2 and then it reaches a plateau of 26% for selectivity above 15. For fixed CO2 concentration in the retentate (i.e., 300 ppm), the stage cut is lower and the CO2 concentration in the permeate is higher for the higher selectivity. As a consequence, the recovery factor which is the combination of these two for fixed feed rate and compositions remains constant. Therefore, the high-selectivity non-porous polymeric membrane with a low surface area is an effective technology to separate CO2from the air to combat global warming