Although significant research efforts regarding new membranes for forward osmosis are underway, less attention has been paid to researching the draw solutions and solutes that could make this technology technically and commercially successful. Preliminary research on one potential draw solute, ethylenediamine tetrapropionic acid (sodium salt) was recently reviewed by Dr. Wang Yan of Huazhong University.
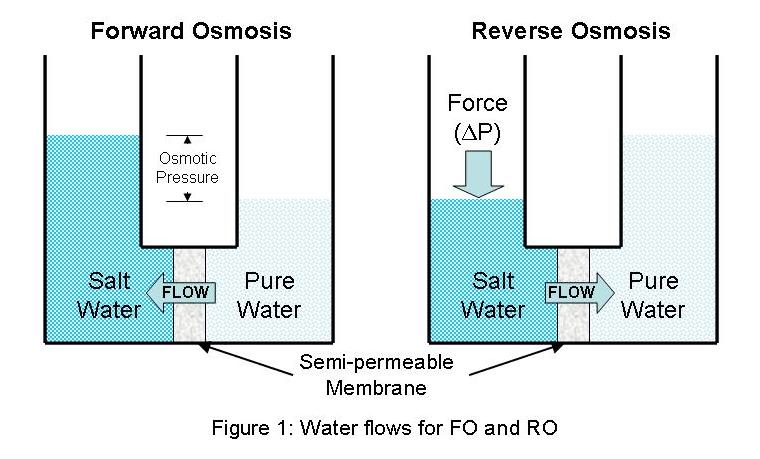
Forward vs. reverse osmosis
Reverse osmosis for water treatment is a multi-billion dollar market worldwide. Both brackish water and seawater treatment provides potable water for huge portions of the population. However, the technology is very capital- and energy- intensive, as the systems and pressures required to overcome the osmotic pressures and produce clean water from these sources are significant. Forward osmosis has recently garnered much investigation due to its relative simplicity and lower energy consumption (the actual osmosis process powers itself), especially in more impoverished and environmentally stressed regions. The most critical aspect of an effective forward osmosis system is the design/choice of the draw solute, especially for systems of continuous production requirements.
Factors for an effective draw solute
Some forward osmosis units that are specific to producing water for human consumption, often for emergency or disaster responses, use a nutrient solution as the draw solute. By design, this nutrient is to be consumed with the water it helps purify, and thus is a "use-once" approach.
In order to use forward osmosis to create general-use potable water, the draw solute is not consumed but ultimately separated from the purified water and recovered in some way. A draw solutes effective for this use:
- Generates a high osmotic pressure, for high water fluxes
- Provides low fouling of the membrane
- Provides high rejection with the membrane chosen to minimize bleed-through losses
- Exhibits a high stability for effective multiple recovery cycles
- Has a low toxicity to minimize the recovery effectiveness sensitivity
Candidates for such draw solutes can include polymers, organic salts, and inorganic salts.
Ethylenediamine tetrapropionic acid (EDTP)
One candidate investigated was the various sodium salts of EDTP: (HOOC-CH2-CH2)2-N-CH2-CH2-N-(CH2-CH2-COOH)2.
EDTP is a stearically large molecule with high solubility. Depending on how many of the four carboxylic acid groups are sodium salts, the pH can easily be varied to test and optimize the properties sought in a draw solute. It is compatible with cellulose triacetate (CTA) membranes across a wide pH range. Preliminary test showed that the optimal balance of flux and salt rejection occurred at pH 8.0, or the 3 Na+ form, with performance significantly greater than simple NaCl in both cases.
The EDTP solute is also stable and doesn't readily agglomerate, which makes it a good candidate for subsequent separation and reuse via filtration technologies such as nanofiltration (NF) and ultrafiltration (UF). In these preliminary tests, NF was the technology of choice.
Equivocal results
Although the flux rates and the salt rejection at around pH 8.0 were good, nanofiltration of the resultant permeate gave only a 70% recovery, owing to the borderline (size-wise) nature of the solute. Having to use NF is also a negative due to its high energy consumption. Additional work with similar molecules but of a higher molecular weight needs to be investigated. If solute recovery can be efficiently achieved with ultrafiltration, with its much lower energy input, the approach may become more economically feasible. There may also be other technologies with better efficiencies than UF/NF.

Dr. Wang Yan