This case study scenario is designed to help students and younger engineers build business problem-solving skills through engineering scenarios. While created to mimic real-life situations, this scenario is hypothetical and for educational purposes only.
Scenario
Our client is one of the world's largest pharmaceutical companies. Based in the US, the client has a wide range of successful products on the markets that include various drugs for erectile dysfunction, lowering blood cholesterol, anxiety disorders, anti-inflammatory drugs, antidepressants, etc. Currently, the client has only one product on the market for cancer treatment generating roughly $500M in annual sales. In the next 6-12 months, the client is planning on introducing a new cancer drug on the market. This particular drug, RFC-9000, will also be for cancer patients, and its target audience is oncologists. In this space, there is one major competitor for the client's current cancer drug. RFC-9000 has already completed clinical trials and is currently awaiting approval from the FDA (the client is expecting FDA approval based upon the trial results). If this new drug is introduced this year, there will be no competition for at least one year. The current sales force includes 750 sales representatives who support the company's cancer drug that is already on the market. To successfully introduce the new drug to the oncology market, the client will need to invest in expanding its sales division.
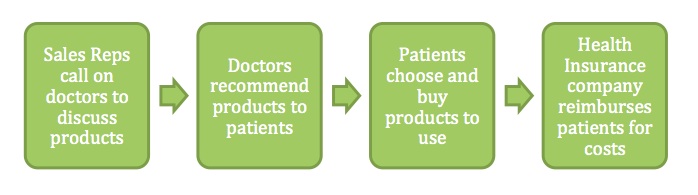
Figure 1: Flow Diagram Illustrating Process of Introducing Pharmaceutical Products to the Customer
The client's intelligence has also suggested that a competitor might introduce a different drug that targets the same cancer within the next 3.5 years.
Your challenge
Your job, as an external consultant, is to help the client think through this problem:
Should the client invest money in expanding its sales force?
Key Points and Assumptions
- The term of patent for a new drug is 20 years but most of that time period is spent in the FDA approval processes. Because of the patent, no generic versions of the drug are expected on the market.
- Being first to market is extremely important to the new product's success.
- Cancer drugs are generally expensive but most of the "out-of-pocket" costs are covered by health insurance companies.
- To sell cancer drugs to oncologists, experienced sales personnel with technical backgrounds will be required with ~6 months to recruit and train new employees.
- FDA approval is not guaranteed; depending on this approval time, new employees might be on overhead until FDA approval is received.



Comments
I'm just a student.. I think the investment needed to come up with a successful drug that has cleared the trials would be so large compared to the hiring of the sales representatives and hence the best decision here is to go forward with the hiring and training of the reps. This is considering the fact that the drug has cleared the clinical trials and just awaiting the final approval. Although not guaranteed, the FDA approval is more likely to be obtained. Also having an FDA approved drug that has no competition in the market and with a potential risk of new drugs from competitors in just under 4 years is a huge risk and could lead to a huge loss in potential revenue.. so again its wise to take some extra risk and invest in the sales force! Any comments/advice would be welcome
Renga, This problem is for students too! As there is no real right answer to this problem, I can say that it also depends on cash on hand to train and hire 750 new sales reps. Let's say for whatever reason, FDA approval couldn't come and the launching was postponed, could the company utilize the new reps in other drugs to still develop the skills they need for RFC-9000 when it hits the market?
tajurg1, I love your response! Just for debate, what happens when a competitor submits their own patent to break the client's patents with a few claims of their own that were for different uses for the drug? - See more at: <a href="http://chenected.aiche.org/tools-techniques/management-case-study-%e2%80%93-pharmaceutical-sales-force-problem/#comments" rel="nofollow">http://chenected.aiche.org/tools-techniques/manag...</a>
Since the management is planning a new product launch, they would have anticipated prior to the launch about the number of packs they plan to sell in the next five years, the intitial budgets that they have, the COGS generated from the 5 year sale plan and the profits generated too. Ofcourse, these are hypothetical figures and estimates, but these will help align the direction they want .. i.e. whether they want to enter the market aggressively or slow. I think an aggressive approach is better if competition threat is anticipated within the next 3.5 years. This will help them establish a brand name by that time. Second, additional sales teams should be hired and trained and pre launch activities can be done in order to maximally utilize resources. Its the synergy between marketing and sales that lies behind all successful product launches.
I like the aggressive approach for pre-launch activities to tell the Oncologists/conferences about the upcoming drug launching. Have you ever seen this not work out though due to technical, supply chain, or development mishaps that forces a delay in the launching?
Hire only cancer survivors who will be able to give testimonies if the drugs helped them.
Not at all client shoild not appoint a new team of sales representative insteadly a client should give this responsiblity to the same team of 750 sales representative because they are already working in market and they also have the knowledge of market and there are aware of there customers if new team is hired it might happen that more time and mone y of the company is invested instead of doing that ideal solution is to give this work to same team as far competitor tensions are concerned when sales team is focused in new product client should modify the old product but saying that the sales team need to sell old product also with the new product such that competitors will not get any idea about the modification as soon as product is modified client should place that product in different divison of his company
Such a nice case study.I would suggest same sales force and give them knowledge about new cancer drug .So that additional cost will not incur and no need to waste time for recruitment and their training also. As available sales force have knowledge about cancer dug and have good contact relation with doctor.Just train with existing sales force and tell them to focus on main advantages over already available cancer drug.While drafting patent ,they need to claim on different uses other than exist and will require IP consultant.
Hi, I am reading your comments and have couple of things to add. I would def. address the following questions/problems: - relation (positive/negative) between the two cancer products. What if a new product results in sales decline of the existing one? There could be a trade off so I would def try to find out more specific details about the target group of the two products, usage etc. - percentage of new sales representative trainings costs of the new product R&D costs - estimation of the period before generics come out - what market share could be covered with existing sales rep thus what is an additional market share (following this profitability) which could be reached by introducing new reps. It seems to me to be pretty financial issue since the question is about investing acctually. Therefore I'd strive to build up kind of sensitivity analysis in order to see whether the investment in new sales representatives makes sense
my poin of view is to debend on the current oncology team in prelunch activity and in the period before recieving the FDA approval that will be suitable due to the follwing 1- that team has a good experiance in the oncolgy sector so you will not invest alot of time in reqrutmient and training 2- you will not have extra expensses or over head untile you have the FDA approval 3- when you have the FDA approval you can expand the no.of feild force
With the cost preposition well-defined, the company might determine their strategy of pricing of the drug. The last step of a go-to-market strategy ought to address the factors of promotion and marketing. Just as many other go to market strategies that are effective, the SHARK Pharma should classically sketch out and use marketing and distribution channels to be able to reach out to the market that is targeted which is the patients with cancer. Before I forget, it should be noted that a go-to market strategy might also be utilized to come up with the imminent SHARK Pharma’s customer relationship administration inventiveness. By observing these rules, Shark Company can be assured that it would manage to introduce the new drug in the market.
From my point of view Since the drug still did no get FDA approval so it means still non-registered items (not yet registered) and based on the compliance the current sales team can not work on it So first of all we need to identify the below points: 1) the Market share for this therapeutic area. 2) the new drug will replace the old drug or each drug will have different patient profile. 3)from the market share we can expect the number of patients might get the benefit of the new drug 4) we can determine the total sales packs for the coming 3.5 years till the competitor come alive 5)the expected price for the new drug. Based on that we can expect the sales value for this product. 6) based on the number of customers list we can expect the number of Med. Rep. require for the new drug or we will utilize the current Med. Rep. 7)Based on the reallocated budget or the cash on hand we can determine the strategic plan for the 3.5 years in terms of selling budget (hiring or utilizing the current Med rep.) and the promotional budget (marketing activities .. launch meetings & RTD ... etc.) Recommendation: since the item non-registered: 1) we can work through the Medical science liaison to cover the top KOL .. filing new patients who can get the benefit from the drug. 2) utilizing the Key account managers to start the inclusion process in the institution & governments hospitals in a collaboration with the MSL. start include the drug as non-registered non-formulary item per patient name based on the patients list created by KOL. 3) Utilizing the regulatory affair department to contact the insurance companies and submit the required documents to start the inclusion plan in the insurance policy. 4) Utilizing the Medical department to conduct different advisory board and pre-launch activities. so an aggressive pre-launch multi-activities from all departments till get the FDA Then once FDA approval got with identifying the above data the plan will follow
As my opinion as the drug has cleared clinical trials and is waiting for fda approval. company can hire 300 sales represantative and train them for 6 months and also 300 represantative from those 750 represantative should be given training for 2 months as they are already in the business. Suppose we get approval we can make these represantative into the business and in the mean time we can hire more professionals .Also our competitor is going to have his drug launch in next 3.5 years . in that time we can create a customer trust and value of drug in patients mind.and if the drug is effective in curing the disease naturally doctors would recommend our drug over the competitors drug to patient.and we can earn margins over a longer period off time.So in my opinion it is better to take risk by hiring those 300 sales represantative and providing perfect training to them.