
.
.Solar collector

His process concept begins with the use of a solar thermal plant incorporating a molten nitrate salt energy medium. This plant provides energy for a pyrolysis reactor as well as for steam generation. The selection of the collector system is driven by the temperatures required (800? C) at the pyrolysis step. With current technology, this requirement can only be met by a power-tower-style collector.
The collector would use underground cement-lined vessels that can hold close to peak thermal energy for more than a week of cloudy days! The energy cost equivalent of this type of system currently ranges from 12-18?/kWhr, but technological advances are expected to reduce costs by 50-65%.
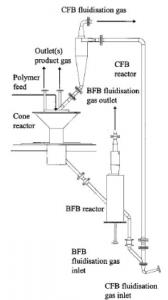
Pyrolysis of waste plastic
Thirty million tons of waste plastics are produced in the United States yearly (2009), with only about 7% being recycled. Disposal by landfill creates land use issues and a loss of value, both material and energy. Incineration avoids the land issue but not the loss of value. Given that recycling typically uses 10% of the energy needed for virgin materials, it makes a prime global energy reduction target.
The preliminary process evaluation protocol led to the selection of a continuous rotating cone pyrolysis reactor, using sand as the heat transfer material. The pyrolysis reaction uses heat to break down the waste plastics into reactive gas products (75-80%). The coke by-product (20-25%) can be utilized for additional energy production.
Carbon dioxide sequestration
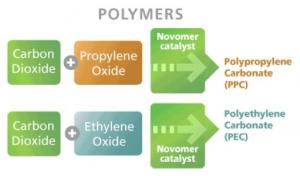

Once the olefins have been produced in the pyrolysis reactor, a catalytic reaction of CO2 and the olefins would then produce polycarbonates. Novomer Inc. is already developing catalysts for various CO2 sequestration reactions. Proprietary catalyst technology has allowed high yields of polycarbonates to be produced from epoxides (ethylene and propylene oxide) and CO2.
The resulting polycarbonates are used for electrical components, data storage and video discs, and in the coatings and construction industries.

Next Steps
The latest development is a concept for a waste plastics recycling process that sequesters carbon dioxide and uses solar thermal energy to remain energy neutral (from a GHG perspective). Next steps indicated as a result of the first evaluation require focus on the development of catalysis and other technologies not yet in existence and a more in-depth understanding and options review of the logistical issues of co-locating the three disparate processes.



Comments
This sounds like a very capital intensive project. I think that there is a lot of uncertainty in the energy market, nothing terribly new except that the source of uncertainty is. I have been listening to a lot of talk lately about the price of natural gas. Falling to prices not seen in decades and really throwing cost calculations into confusion. Depending on where the price settles - there could be a significant restructuring of energy cost basis. But that price also depends on infrastructure changes such as retro-fitting power plants to move from coal to gas. I guess what I am saying is that it would be a difficult time to be a decision maker trying to predict the cost-competition a few years out. Though this project has a tremendous benefit of being tied into other valued processes. It could provide enough reinforcement to get it started.