
Dr. Bruce Hinds and his team at the University of Kentucky are exploring the properties and performance of carbon nano-tubes (CNTs) for water treatment applications. By exploiting the inherent flow properties of the tube interior, creating "gatekeeper" functionality at the entrances and exits, and allowing for "back side" signaling, precisely engineered separations membranes that mimic the precision of biological membranes are possible
The Carbon Nano-Tube Membrane Concept
Think of a single molecular thick sheet of graphite carbon atoms on a hexagonal grid. Now, take that sheet
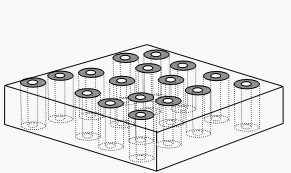
and roll it into a cylinder, and you have the concept of a CNT. If you then make bundle sheets of these tubes, and via several tricky steps of stoppering the tubes, casting the lot in polymer, etching away excess to expose the ends, then un-stoppering, you can create a CNT membrane sheet. Current tube diameters in lab testing are around 7 nm ID and 40 nm OD.
A Nanoscopic Slip 'n' Slide
Inside tube surface is an "anatomically flat" hydrophobic core, with large Van der Waals distances, which become important in the fluid transport properties. The smooth tube surface yields virtually no back-scatter, and with its hydrophobic nature, provides a condition of "perfect slip" for water molecules flowing in the tube. Flow velocities that are tens of thousands of times faster than through similarly sized (diameter) pores are achieved. Flow velocities of 10-40 cm/s have been demonstrated.
Chemical Functionalization: Key Masters and Gatekeepers
In the process of making the CNTs, the exposed end carbon atoms become carboxylated. This is very convenient in that it allows for the addition of many different chemical moieties to the entrance and exit of the tube, and thus that can affect how rapidly, or even if, solutes can enter the tubes.
Long polymer chains attached to the ends can inhibit flow into the tube. As the CNTs are conductive, you can apply a charge potential to the surface which restricts flow even more so (think of what rubbing a balloon on your head does to your hair). Flux is found to increase when charged molecules are placed at entrance. To date, a "gatekeeper" that would allow CNT effective use as a desalination membrane has not been discovered. This may also be a result of the "large" ID of the current CNTs in testing.
Next Steps
There is still much work to do for desalination effectiveness, including incorporating very high charge density at the inlet end of the tubes, plus making tubes with much smaller diameters, on the order of 0.8 nm.

Other key challenges include:
- Porosity improvements: a high percentage of the tubes in the membrane are or become blocked.
- Good scale-up: these are "lab-made" materials that will need to be mass produced.
- Appropriate ion exclusion properties for "Desal" type applications
- Solve the observed concentration polarization issues.


