2022 Annual Meeting
(59a) Controlling the Immune Response through Controlling the Aggregation Kinetics of Engineered Peptides
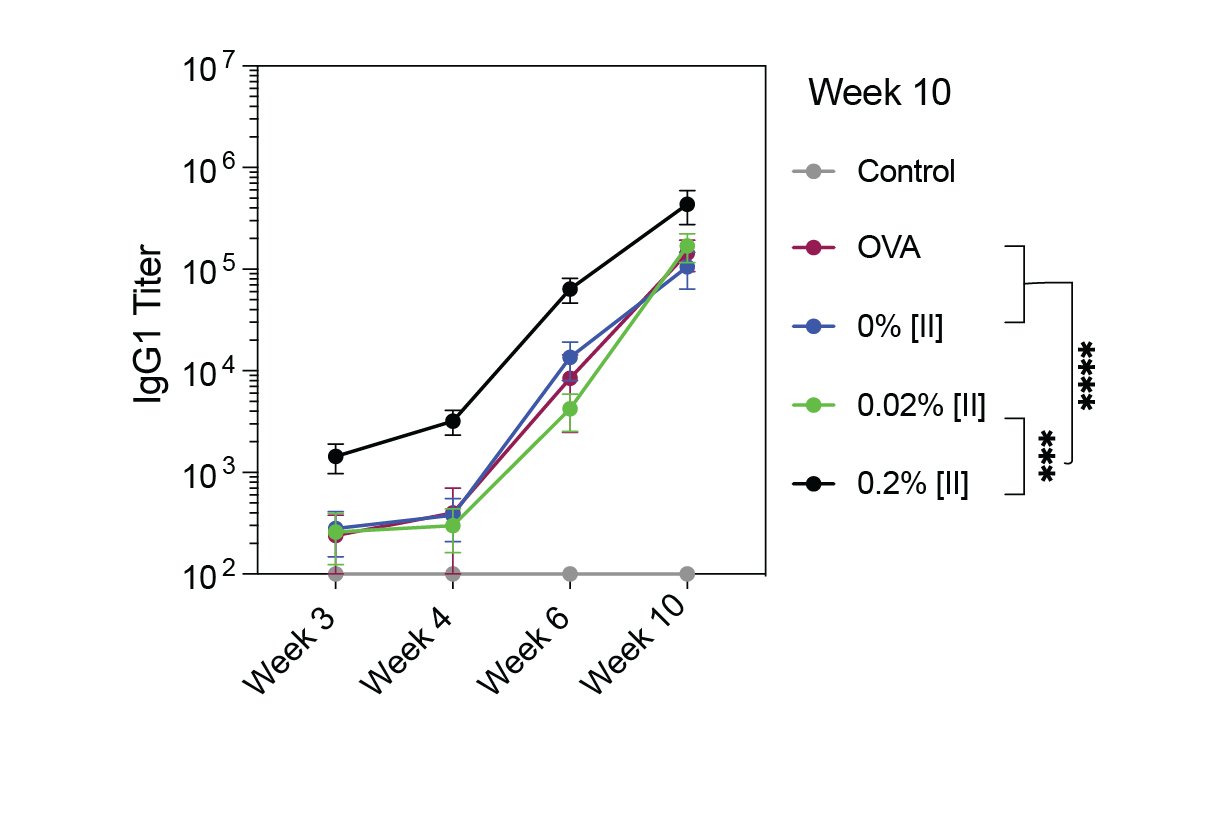
In this study, we showed the control over peptide assembly, as in Aβ1-42 oligomerization, to control the immune response effect of PAIIR, as vaccine adjuvants.2 In this study, we created aggregates of the PAIIR that can assemble in different kinetics (shown as 0.002% fastest and 0.2% slowest). The aggregation kinetics affected their cell membrane rupture kinetics, thus their immunogenicity. We showed detailed characterization of the peptide aggregation, membrane damage, and cell death profiles of these aggregates on several cell lines. We used ovalbumin (OVA) as the antigen and prepared cocktails of different aggregation PAIIRs. Antibody formation in mice followed the same aggregation kinetics profile (Fig.1); the slowest aggregation provided the highest number of antibodies. In this study, we showed controlling the effects of biomaterials on the cells and overall organism, through molecular engineering principles.
[1] Hamsici, S. et al. Peptide framework for screening the effects of amino acids on assembly. Science Advances (2022)
[2] Gunay, G. et al. Peptide Aggregation Induced Immunogenic Rupture (PAIIR). BioRxiv (2022)