2022 Annual Meeting
(478f) Tuning the Electronic Metal-Support Interaction to Design Stable and Active Oxygen Reduction Electrocatalysts
Authors
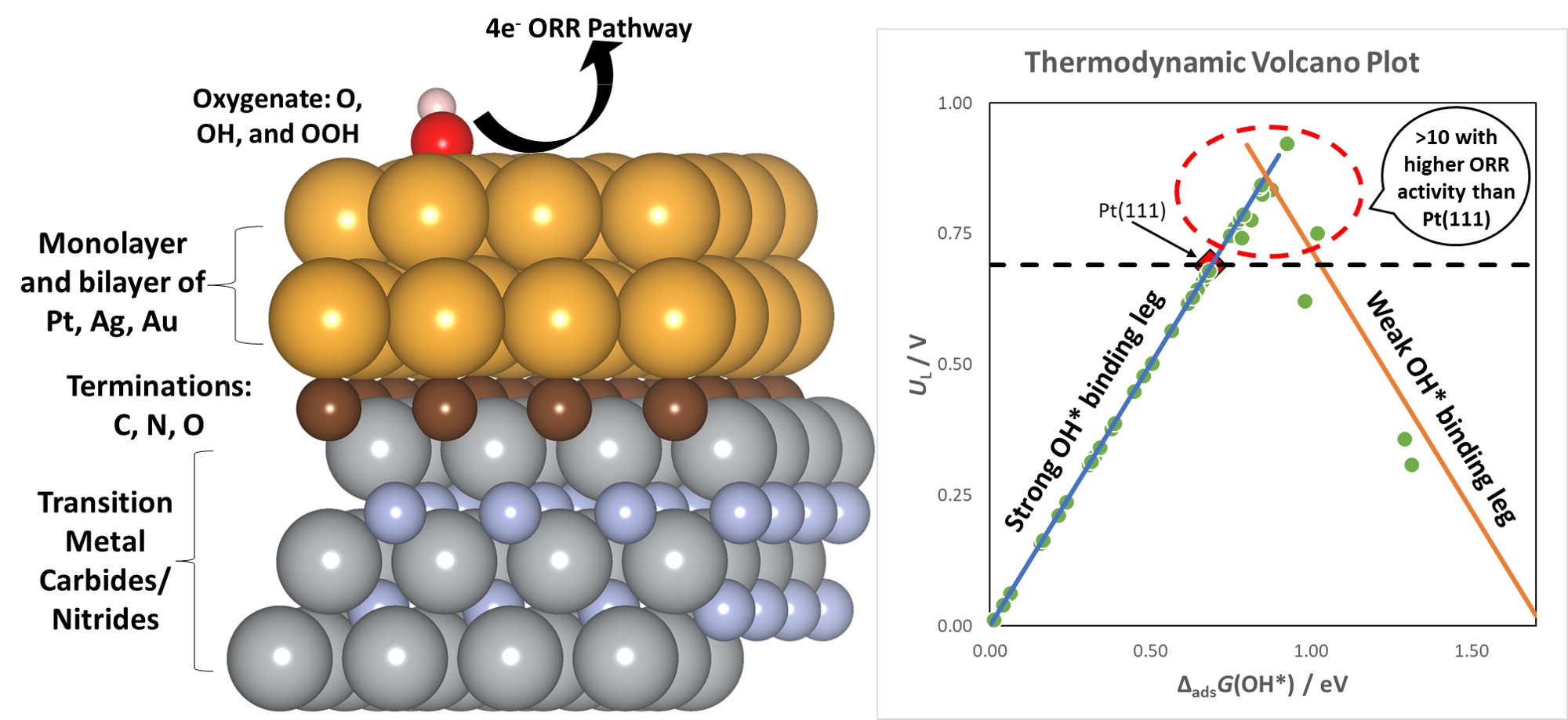
Using first principles methods, we design heterostructures containing monolayer and bilayer metal films supported on the hexagonal facets of carbides and nitrides. These supports are terminated by O*, C*, N*, and the bare metal. The thermodynamic and electrochemical stabilities of the films are first assessed. The computational hydrogen electrode (CHE) is adopted to evaluate the catalytic activity of ORR on the stable supported films by explicitly calculating the limiting potentials (UL). A micro-solvation model is employed to increase the accuracy of the UL calculated.
We construct a volcano plot correlating to the free energy of adsorption of OH*. Upon screening > 200 heterostructures, we find at least 10 metal/support combinations that are more active than Pt(111). Due to the exquisitely tuneable EMSI, the pool of materials at the volcano maximum is widened, with several monolayered films of Ag and Au demonstrating higher activity than Pt(111). The EMSI modifies the scaling relations amongst the oxygenates on supported metal films as compared to the fcc(111) surfaces of the PGMs. These modified scaling relations result in a higher theoretical maximum for . We show that the pool of heterostructures that demonstrate high activity for oxygen reduction also possess lower overpotentials for oxygen evolution, thus hinting that metal films supported on carbides/nitrides are promising bifunctional electrocatalysts.