2022 Annual Meeting
(390c) Connection between Local Dynamic Environments & Relaxation Mechanism
Authors
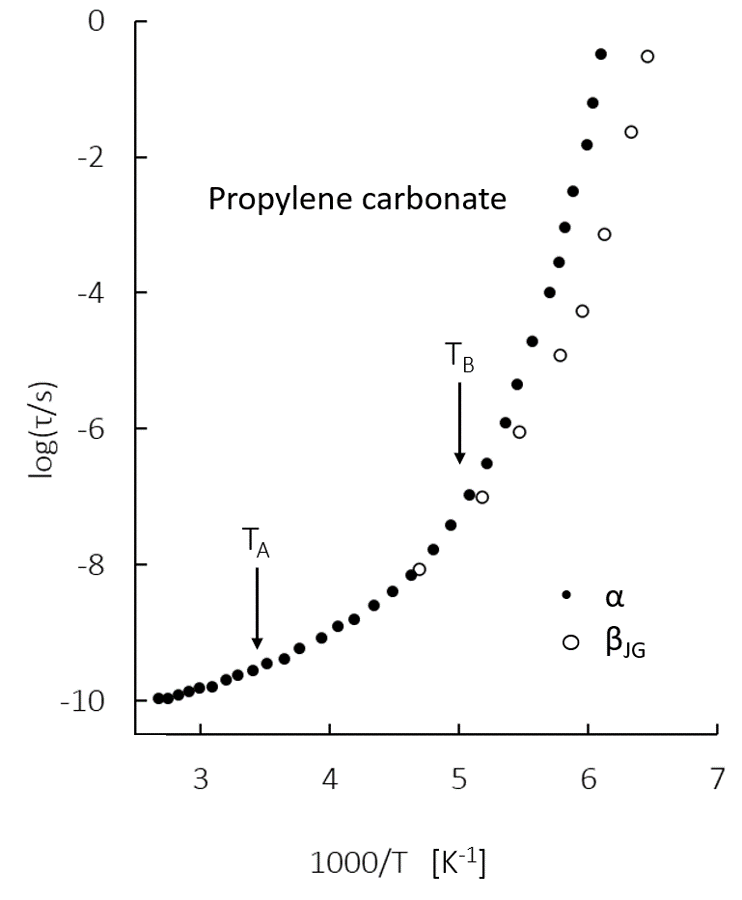
As shown in the figure1, in the liquid and glass phases, there are temperatures TA and TB at which mechanistic changes in relaxation occur2. At relatively high temperatures, T > TA, relaxation is purely collisional and there is no evidence of cooperative motion, while at intermediate temperatures TB > T > TA, dynamics follow super-Arrhenius behavior. At low temperatures, T < TB, a secondary relaxation process that bifurcates from primary relaxation emerges. Using data collected from neutron scattering of five simple liquids3, our group has seen that at TA and TB, there is a shift in the average number of excitations in the first shell of the molecule. Further, we can categorize local dynamic environments based on the number of excitations in the first shell; these environments are titled Piwhere i denotes the number of excitations in the first shell. By assuming a random distribution of these environments, we have seen that at temperatures TA and TB, environments P1 and Pâ¥2 both have populations of approximately 24%, respectively. These thresholds for excitations paint a fundamental picture of the local processes lead to the mechanistic changes in liquid relaxation. With this information, we reach closer a level of predictive frameworks for liquids comparable to that we have for solids and gases.
- Schneider et al., Phys Rev E59 (1999)
- Stickel et al., J Chem Phys 104, 2043 (1996)
- Cicerone et al., Phys Rev Lett 113, 117801 (2014)