2022 Annual Meeting
(357g) Development and Evaluation of Data-Driven Control Strategies for Drying End-Point Determination in a Semi-Continuous Fluid Bed Granulation Process
Authors
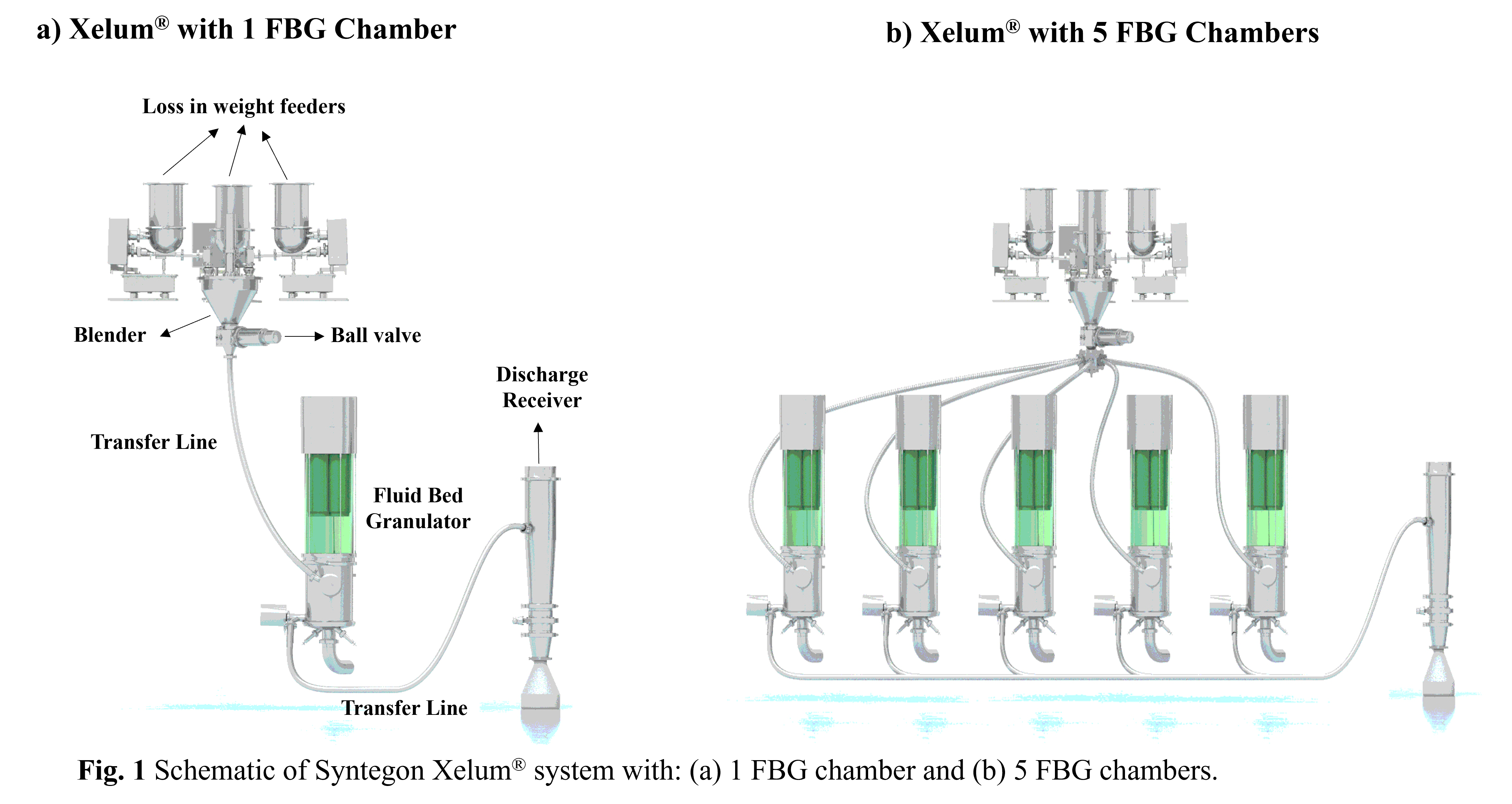
In an FBG process, residual moisture content is an important intermediate quality attribute requiring a robust monitoring and control strategy for it to be maintained within pre-defined bounds. Industrially relevant approaches for predicting the moisture content in granules have mainly revolved around mechanistic heat and mass balances. While these approaches have yielded encouraging results, the use of such models has been mostly limited to technology transfer, design space exploration, and deviation investigations. Since such methods may require expert knowledge and more involved validation and maintenance strategies, their use in routine manufacturing is often limited. Another common approach has been to utilize at-line or in-line Near-infrared (NIR) spectroscopy-based tools to predict the moisture content in granules. While at-line NIR involves physical granules sampling, in-line NIR may suffer from probe fouling (exacerbated by the presence of binder liquids). Implementation of NIR tools in routine manufacturing also involves time and resource intensive model calibration, validation, and subsequent maintenance at the manufacturing site, which makes integrating in-line NIR in the 5-chamber unit (i.e., one probe per chamber) cumbersome. Thus, there is a need for intuitive, easy to use, and robust practice-based drying end-point approaches. Additionally, such approaches should enable easy maintenance with periodic validation using in-process loss-on-drying measurements and system calibration.
The present work aims to develop and evaluate two control strategies for drying end-point determination of a FBG process to ensure targeted and repeatable residual moisture contents of the discharged granules. Both strategies utilize logged process variables (measured by the process sensors) that reflect the process state based on established drying principles for porous granules. In the first strategy, one-sided inequality constraints were established for the process variables (bed temperature, outlet air absolute humidity & temperature) while incorporating measurement noise. The second strategy involved tracking two aspects of the outlet air humidity during drying: (a) difference between the absolute humidities of the outlet & inlet air and (b) change in gradient of the outlet air absolute humidity. The absolute humidity difference between the outlet and inlet air ensured that the drying process is at or beyond the falling rate drying period after which the outlet air humidity gradient was tracked within pre-defined bounds (near zero), thus indicating drying completion. The specifications for both the strategies were established based on several granulated sub-lots where both formulation properties and process conditions were varied. The performance of the developed strategies was assessed on an independent extended development campaign, which confirmed that the proposed data-driven approaches can be leveraged as drying end-point control strategies in routine manufacturing.
Research Interests - Continuous powder processing, novel PAT application for enhanced process understanding, microstructure characterization, and novel small molecules based drug delivery systems.