2022 Annual Meeting
(172c) A Mathematical Model to Predict the Drug Release Profile in Bilayered Osmotic Controlled Release Tablets
Authors
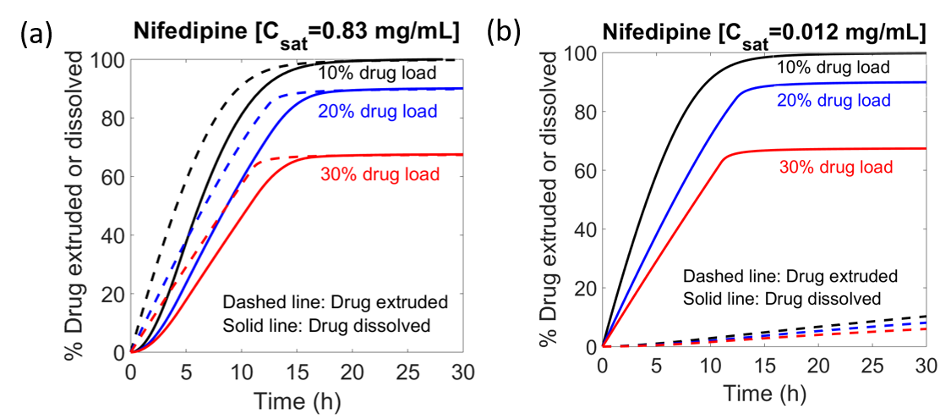
In this work, we model a bilayer osmotic controlled-release tablet to predict the drug release rate as a function of several key parameters. These parameters include the coating thickness and the tablet size as well as the ratios of excipients and active pharmaceutical ingredient. The model describes dynamically all the main release processes occurring during the dissolution of the tablet, namely, the solvent influx driven by the difference in osmotic pressure across the coating in both drug and sweller layer, dispersion of core components (drug and polymer), swelling of the tablet due to solvent accumulation, build-up of hydrostatic pressure inside the tablet, tensile stress acting on the coating, and the extrusion of the dispersed core components, dissolution of drug particles in bulk media, swelling of the polymer in the sweller layer, and pressure exerted by sweller layer onto the drug layer. The model is validated by comparing the predictions with drug release data for two drugs of varying solubility and for the same drug in different dissolution media. The drug release predictions match the measurements well, confirming that our model is well suited for elucidating the osmotically controlled drug release process in SCT tablets.
The attached figure presents the model predictions of drug release profile for the same drug but in two different dissolution media with different solubility. The figure plots (a) model predictions of drug release in dissolution media with high solubility, and (b) model predictions of drug release in water having low drug solubility. Presence of the sweller layer ensures that the drugs are extruded out at the same rate despite the large variation in solubility.