2020 Virtual AIChE Annual Meeting
(705d) Generating Acidity in Metal Oxides with SiO2 Overcoats
Current synthesis techniques make it difficult to independently tune these important parameters. For example when making zeolites through hydrothermal routes, small changes to the synthesis solution can significantly impact crystallite size potentially creating mass transfer limitations. Similarly, the properties of amorphous silica-alumina are highly dependent on the synthesis procedure followed. It is our aim to employ techniques derived from supported oxide catalyst synthesis to precisely control acid site distribution, strength, and density in mixed oxides.
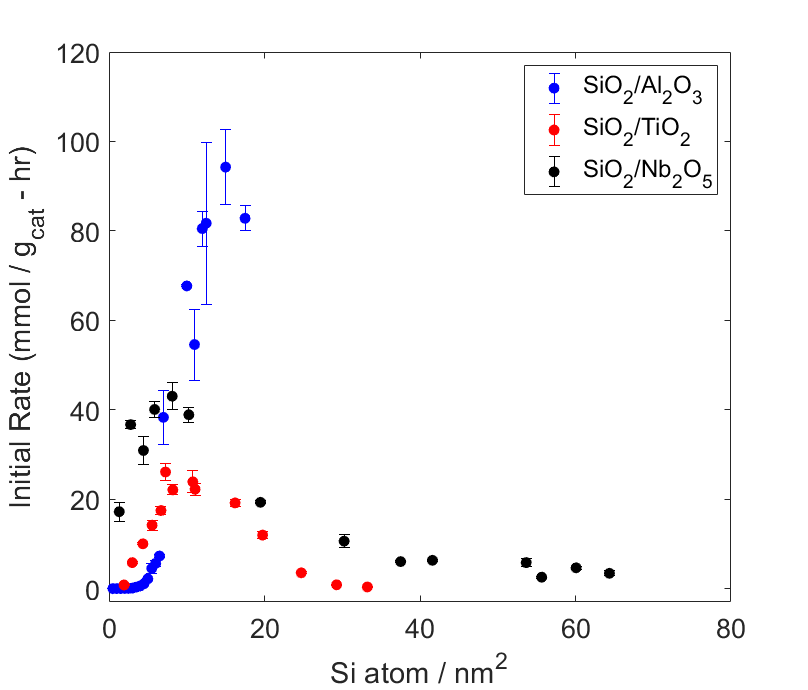
In this work we investigate the properties of acid sites generated upon the grafting of SiO2 onto Nb2O5, TiO2, and Al2O3. For each metal oxide support, a series of catalysts with varying surface Si densities were prepared using a previously reported cyclic sol-gel process followed by thermal treatment in static air. The acid properties of the prepared catalysts were studied using pyridine DRIFTS, NH3 TPD, and propylamine TPRx.
The reactivity of the SiO2/MOx catalysts was probed by the liquid-phase screening reaction of 3,4-dihydropyran with n-octanol. Reactions run at 70ºC show that the initial reaction rate increases with increasing surface Si density to a maximum before sharply decreasing for each of the SiO2/MOx series tested. Preliminary characterization of these materials indicates that their acid properties and activity are strongly dependent on the surface Si density, which can be precisely controlled through the synthesis we have employed. Current work is aimed at extending this insight to engineer acid catalysts for light alkene oligomerization.