2020 Virtual AIChE Annual Meeting
(3p) Exploring Artificial Photosynthesis By Synthetic Biology and Protein Engineering
Author
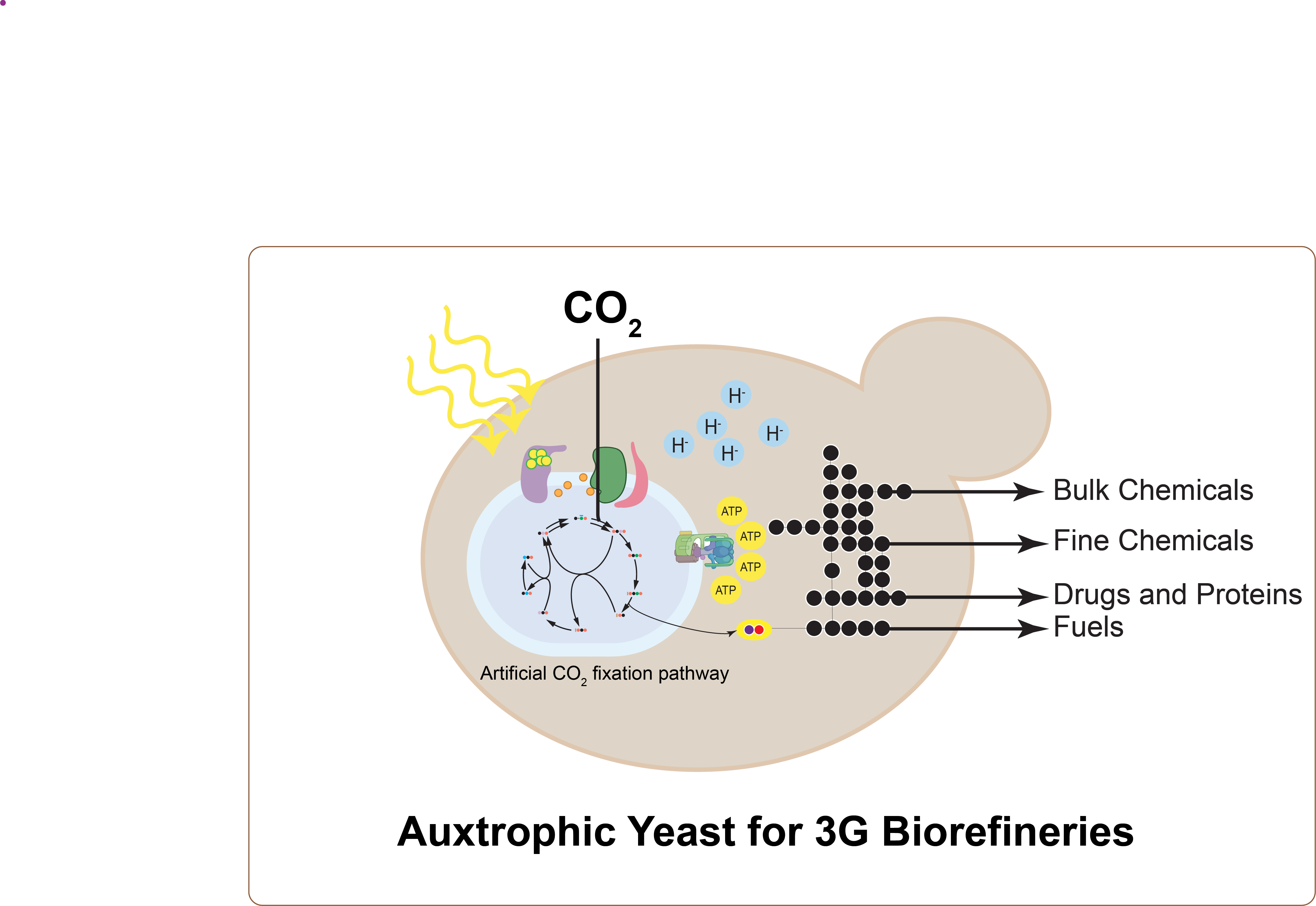
Continuous petroleum depletion and global warming have spurred interest in renewable alternatives to fossil fuels. Biotechnology provides environmentally friendly alternatives to produce fuels and chemicals in a carbon-neutral manner1. However, current bioproduction processes mainly use sugar-based feedstock (the first generation) and biomass (the second generation) and suffer from low energy conversion efficiencies and low productivities. Switching from the organic feedstock to the atmospheric CO2 (the third generation-3G) is one of the most important targets to meet the requirement of sustainable development2. To fulfil the goal of 3G biorefineries, converting CO2 to value-added chemicals, there have been a lot of efforts to engineer autotrophs to accommodate heterologous production of bioproducts3â5. Further improving CO2 and energy utilization efficiency and expanding repertoire and productivity of chemicals that can be generated from autotrophies are two major bottlenecks in 3G biorefineries1. As an alternative, popular heterotrophic microbial cell factories such as E. coli and yeast have been successfully engineered to include functional CO2 fixation pathways most recently6,7. Thanks to comprehensive knowledge over E. coli and yeast together with ready-to-use metabolic network for production of various chemicals and advanced metabolic engineering and synthetic biology tools8,9, there are great opportunities to convert yeast to become efficient 3G microbial factories.
My research goal is to develop protein engineering and synthetic biology platforms to construct and engineer CO2 fixation pathways and artificial light-reaction of photosynthesis in heterotrophs, especially in yeast (S. cerevisiae and P. pastoris). Yeast is one of the most prominent microorganisms used for production of biofuels, fine chemicals, pharmaceuticals, and enzymes10â12. As a versatile model eukaryotic organism, yeast has been used to express functional genes from different hosts, both prokaryotes and eukaryotes, and the Calvin-Benson-Bassham (CBB) cycle has been successfully constructed in Pichia pastoris 6. Especially due to the availability of well-developed engineering tools in yeast, it is highly feasible to engineer yeast to become an efficient photoautotrophic organism converting CO2 to useful products. In addition, yeast could serve as a platform to enable us to understand and engineer photosynthesis to certain level that could not be achieved in higher eukaryotes such as algae and plants. My future research will integrate bioinformatics, synthetic biology, metabolic engineering, protein engineering and computational biology to (1) engineer artificial CO2 fixation pathways in yeast; (2) engineer artificial microbes to produce chemicals from solar energy, and (3) study and engineer phototrophic light-dependent reactions in yeast.
References:
(1) Liu, Z.; Wang, K.; Chen, Y.; Tan, T.; Nielsen, J. Third-Generation Biorefineries as the Means to Produce Fuels and Chemicals from CO 2. Nature Catalysis 2020, 3 (3), 274â288.
(2) Musa, S. D.; Zhonghua, T.; Ibrahim, A. O.; Habib, M. Chinaâs Energy Status: A Critical Look at Fossils and Renewable Options. Renewable and Sustainable Energy Reviews 2018, 81, 2281â2290.
(3) Liao, J. C.; Mi, L.; Pontrelli, S.; Luo, S. Fuelling the Future: Microbial Engineering for the Production of Sustainable Biofuels. Nature Reviews Microbiology 2016, 14 (5), 288â304.
(4) Carroll, A. L.; Case, A. E.; Zhang, A.; Atsumi, S. Metabolic Engineering Tools in Model Cyanobacteria. Metabolic Engineering 2018, 50, 47â56.
(5) Banerjee, C.; Dubey, K. K.; Shukla, P. Metabolic Engineering of Microalgal Based Biofuel Production: Prospects and Challenges. Front. Microbiol. 2016, 7.
(6) Gassler, T.; Sauer, M.; Gasser, B.; Egermeier, M.; Troyer, C.; Causon, T.; Hann, S.; Mattanovich, D.; Steiger, M. G. The Industrial Yeast Pichia Pastoris Is Converted from a Heterotroph into an Autotroph Capable of Growth on CO 2. Nature Biotechnology 2020, 38 (2), 210â216.