(724d) Catalytic Conversion of CO2 into Vinyl Acetate with Activated Carbon Based Zn Acetate Catalyst
AIChE Annual Meeting
2020
2020 Virtual AIChE Annual Meeting
Fuels and Petrochemicals Division
Value-Added Chemicals from Natural Gas
Friday, November 20, 2020 - 8:45am to 9:00am
CO2 + CH4 → CH3COOH; (2) CH3COOH + C2H2 → vinyl acetate
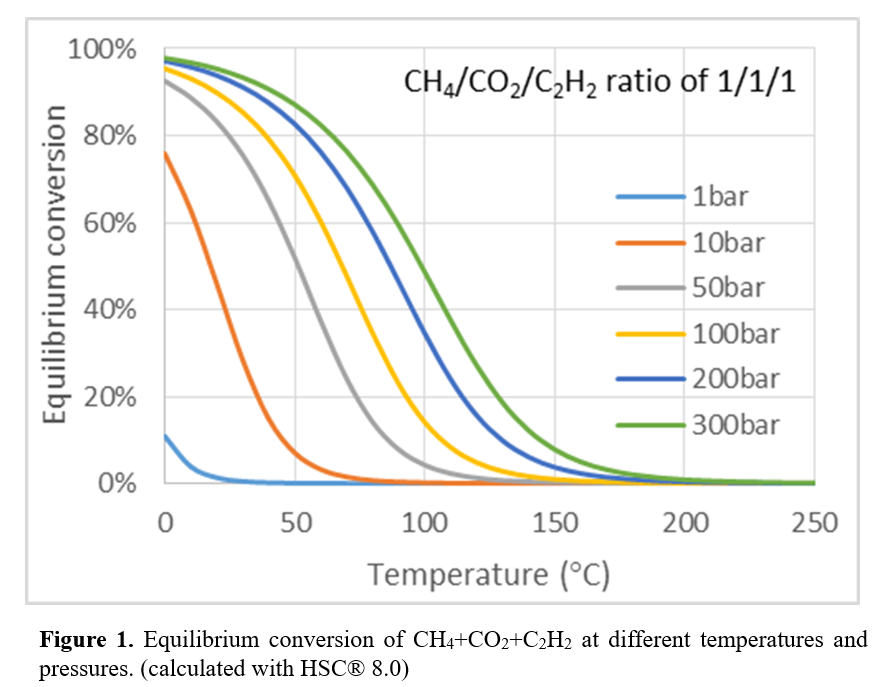
This formation of acetic acid is thermodynamically limited, but can be addressed by coupling this reaction with a thermodynamically favorable reaction, e.g., reacting acetic acid with acetylene, and forming vinyl acetate monomer (VAM), a high-value industrial intermediate. The overall coupled reaction with CH4 + CO2 + C2H2 to VAM is thermodynamically feasible (ΔGro= +6.4 kJ/mol). Previously, Wilcox et al. demonstrated the catalytic reaction of VAM from this reaction at temperature as high as 4000C under atmospheric pressure [1], but VAM formation was not stable at these conditions (Figure 1). The formation of VAM from CH4 + CO2 + C2H2 involves one, single reaction at low temperature and high pressure. The goal of this work is to investigate and synthesize an active and stable catalyst at thermodynamically favorable reaction conditions. Keeping this in mind, activated carbon based zinc acetate (ZnA/C) was synthesized and tested at different reaction conditions.
After characterizing ZnA/C with SEM-EDS, Raman and XRD, VAM reaction was run from temperatures as low as 1300C and at a pressure of 9 bar for ~15 hours. A stable VAM formation was observed. Acetaldehyde was observed as a side product of VAM. When the temperature was increased to 1600C, VAM Selectivity increased as well. VAM selectivity also increased with lower space velocity. Activity decreased slightly with time. Catalytic deactivation can be observed with the loss of acetate from the catalytic surface at high temperature.