(582e) Coupled NO and Hydrocarbon Trapping, Release and Conversion on Pd/SSZ-13 and Pt-Pd/BEA
AIChE Annual Meeting
2020
2020 Virtual AIChE Annual Meeting
Catalysis and Reaction Engineering Division
Emissions Control I: Passive NOx Adsorbers and Hydrocarbon Traps
Wednesday, November 18, 2020 - 9:00am to 9:15am
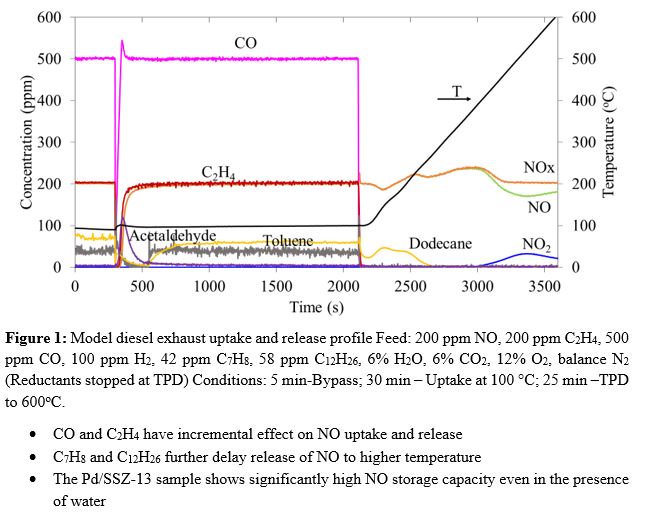
Vehicular emissions control technologies such as the three-way catalyst (TWC) for gasoline and the diesel oxidation catalyst (DOC) + selective catalytic reduction (SCR) for diesel are largely effective in eliminating CO, hydrocarbons (HCs), and NOx above 200°C. However, emission control during vehicle cold-start (< 200ºC), remains a major challenge across all vehicle types. The challenge is exacerbated by the lower exhaust temperatures and higher HC concentrations of advanced lean-burn engines. To mitigate these cold-start emissions, NOx and hydrocarbons may be trapped on selective adsorbents at low temperature and released/converted at higher temperature. In this study the effectiveness of coupled NOx and HC trapping and conversion on a model Passive NOx Adsorber (PNA) catalyst (Pd/SSZ-13) and a hydrocarbon catalytic trap (Pt-Pd/BEA) is examined. Uptake and release experiments are conducted over a wide range of operating conditions to assess the effectiveness and synergy of the materials. Figure 1 shows the outcome of the model diesel exhaust during a simulated uptake and release experiment on Pd/SSZ13. Nearly complete uptake of NO is achieved at 100 °C for about 50s. Experiments show that common exhaust components C2H4 and CO have an incremental benefit on NO uptake. It is found that NO and CO/C2H4 uptake are mutually inhibitive due to competition for adsorption on the zeolitic Brønsted acid sites (BAS). While CO/C2H4 inhibit NO adsorption on BAS, they promote NO storage on Pd and beneficially delay NO desorption to higher temperatures. Similar experiments with the HCT will shed more light on the HC uptake, release, and oxidation. Ongoing experiments using the combined PNA and HC trap materials are underway to improve the performance towards the optimum design of multi-functional trap. Conditions are identified for which > 90% NOx trapping is achieved as a first step towards developing multi-functional materials for coupled NOx and HC trapping/conversion.