(25e) Solubility and Nucleation Kinetics of a Series of Representative Single and Mixed Diesel N-Alkanes in Dodecane and Toluene
AIChE Annual Meeting
2020
2020 Virtual AIChE Annual Meeting
Particle Technology Forum
Particles, Crystals and Agglomerate Formation Dynamics and Modeling
Monday, November 16, 2020 - 9:00am to 9:15am
Current wax mitigation strategies involve the development of chemical additives to control or inhibit the nucleation and growth of crystals so that they no longer form large deposits or block filters. However, the variable nature of fuel composition, specifically the fractionation of the n-alkanes, results in a complex challenge for the additive provider to meet operational needs1. In order to understand crystallisation of waxes in fuels, it is important to understand the crystallisation of n-alkanes in mixtures and singularly.
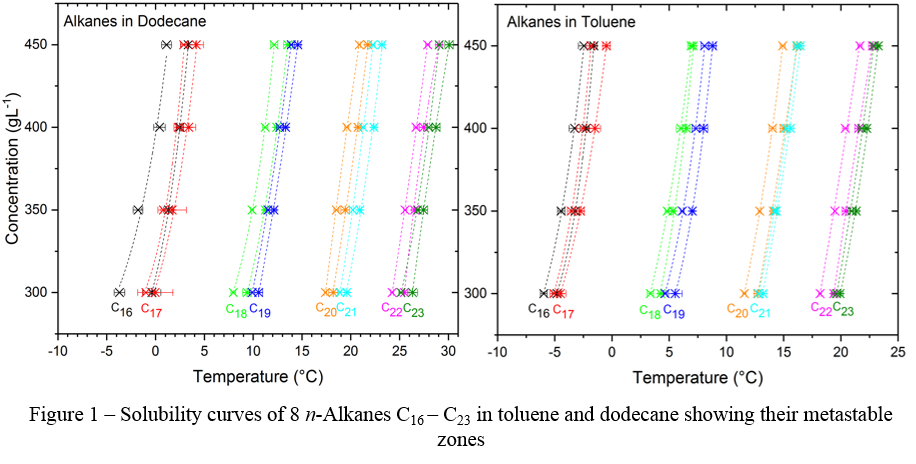
Here we present the results from an experimental study on the nucleation kinetics and solubility behaviour single and mixtures of 8 n-alkanes representative of diesel fuel, ranging from C16H34 to C23H48 in toluene and dodecane. Analysis of the experimental polythermal data making, use of the analytically derived KBHR approach2–4 reveals, that in all bar two cases the preferred nucleation mechanism for single n-alkane solutions is instantaneous. i.e. all the nuclei are formed at once at a certain level of supersaturation, to further grow . Furthermore, the influence of chain length on solubility was observed between odd and even alkanes in both toluene and dodecane. By analysing the nucleation and solubility behaviour within the set of n-alkanes tested, suggestions of the factors controlling wax crystallisation in fuel systems are presented and discussed from first thermodynamic principles.
References
[1] A. R. Gerson; K. J. Roberts; J. N. Sherwood, Powder Technol., 65 (1991) 243.
[2] D. Kashchiev; A. Borissova; R. B. Hammond; K. J. Roberts, J. Cryst. Growth, 312 (2010) 698.
[3] D. Kashchiev; A. Borissova; R. B. Hammond; K. J. Roberts, J. Phys. Chem., 114 (2010) 5441.
[4] D. M. Camacho Corzo; A. Borissova; R. B. Hammond; D. Kashchiev; K. J. Roberts; K. Lewtas; I. More, CrystEngComm, 16 (2014) 974.